Team:SDU-Denmark/Tour40
From 2014.igem.org
| Line 43: | Line 43: | ||
<h4>Flavor improvement</h4> | <h4>Flavor improvement</h4> | ||
<p> | <p> | ||
| - | <span class="intro">The thought of eating <i>E. coli</i></span> does not sound that delicious – and that is why we want our OneProt to taste like lemon. | + | <span class="intro">The thought of eating <i>E. coli</i></span> does not sound that delicious – and that is why we want our OneProt to taste like lemon. Though the cloning proved unsuccessful. However, we have characterised an odor-free <i>E. coli</i> strain and compared it with a wild type K12 MG1655 <i>E. coli</i> strain. We have used Ion Mobility Spectroscopy to analyse the two strains, since we deemed it among the best methods to analyse odors. From the results it was shown that the odor-free strain does not produce indole, which is the compound responsible for the characteristic door of <i>E. coli</i>. |
<br><br> | <br><br> | ||
</p> | </p> | ||
Revision as of 23:59, 17 October 2014
Results
"I know that I am intelligent, because I know that I know nothing." – Socrates
The next few pages will guide you through the results of the characterization of our submitted parts. On this page, you will find short descriptions of our results, leading you to the final result. We hope you will dig deeper into the results of our systems. After the need to prioritize subprojects, the aim of our project was to get E. coli to express a self-designed nutritional protein, controlled by an inducible promoter. Furthermore it will express limonene synthase for the synthesis of limonene, the main part of lemon flavor.
Synthesis of self-designed protein OneProt
Pictures of worms/WB
Growth curve
Controlling the Tet promoter
 Figure 2: Plate containing 200 ng/mL doxycycline plated with E. coli WT and E. coli expressing GFP controlled by a constitutive promoter, pTet-TeR(+LVA) and pTet-TetR(no LVA).
Figure 2: Plate containing 200 ng/mL doxycycline plated with E. coli WT and E. coli expressing GFP controlled by a constitutive promoter, pTet-TeR(+LVA) and pTet-TetR(no LVA).
 Figure 1: Plate containing 0 ng/mL doxycycline plated with E. coli WT and E. coli expressing GFP controlled by a constitutive promoter, pTet-TeR(+LVA) and pTet-TetR(no LVA).
In order for us to be able to control the amount of OneProt produced, we need a promoter that can be controlled. For this, we put OneProt under expressional control by pTet. Besides investigating the controllable expression, we also investigated the influence of the LVA tag on TetR. We proved that the inhibition of pTet by TetR is correlated with the concentration of inducer – increasing amounts of inducer means decreasing levels of inhibition of pTet. Comparing the expression by pTet controlled by TetR with and without LVA tag shows that there is a higher expression upon induction, using TetR with LVA than using TetR without LVA. We also show that the pTet is leaky.
Figure 1: Plate containing 0 ng/mL doxycycline plated with E. coli WT and E. coli expressing GFP controlled by a constitutive promoter, pTet-TeR(+LVA) and pTet-TetR(no LVA).
In order for us to be able to control the amount of OneProt produced, we need a promoter that can be controlled. For this, we put OneProt under expressional control by pTet. Besides investigating the controllable expression, we also investigated the influence of the LVA tag on TetR. We proved that the inhibition of pTet by TetR is correlated with the concentration of inducer – increasing amounts of inducer means decreasing levels of inhibition of pTet. Comparing the expression by pTet controlled by TetR with and without LVA tag shows that there is a higher expression upon induction, using TetR with LVA than using TetR without LVA. We also show that the pTet is leaky.
Flavor improvement
The thought of eating E. coli does not sound that delicious – and that is why we want our OneProt to taste like lemon. Though the cloning proved unsuccessful. However, we have characterised an odor-free E. coli strain and compared it with a wild type K12 MG1655 E. coli strain. We have used Ion Mobility Spectroscopy to analyse the two strains, since we deemed it among the best methods to analyse odors. From the results it was shown that the odor-free strain does not produce indole, which is the compound responsible for the characteristic door of E. coli.
Added parts and devices
To the great iGEM Registry of Standard Biological Parts we have added 3 basic parts, 2 regulatory devices and 4 regulable production devices.
 "
"

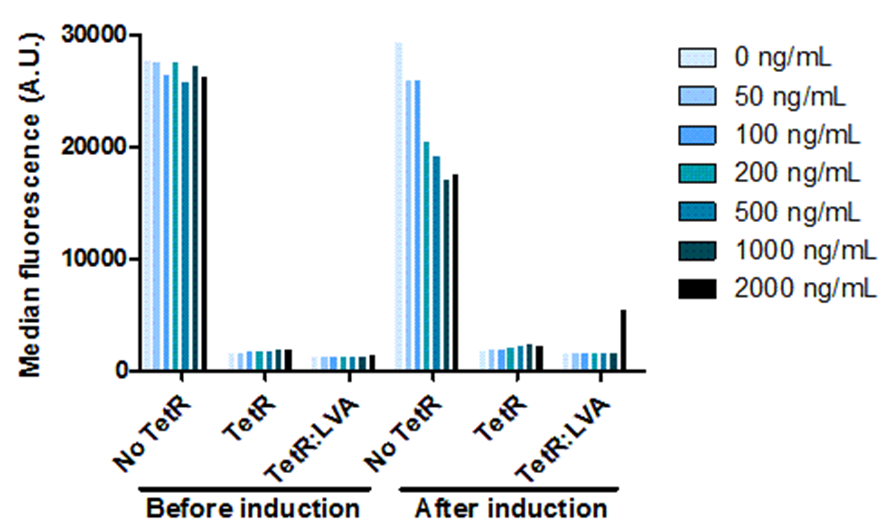 Figure 3: Dose response to doxycycline.
Figure 3: Dose response to doxycycline.