Team:Paris Saclay/Notebook/August/22
From 2014.igem.org
(Difference between revisions)
(→D - Lemon Scent) |
m (→pPSI dephosphorylation) |
||
| (30 intermediate revisions not shown) | |||
| Line 2: | Line 2: | ||
=Friday 22nd August= | =Friday 22nd August= | ||
==Lab work== | ==Lab work== | ||
| - | === | + | ===Lemon Scent=== |
====pPSI digestion==== | ====pPSI digestion==== | ||
| - | |||
| - | + | {| class="wikitable centre" width="25%" | |
| - | + | |+ | |
| - | + | |- | |
| - | + | ! scope=col | components | |
| - | + | ! scope=col | volumes | |
| + | |- | ||
| + | |pPSI | ||
| + | |40μl | ||
| + | |- | ||
| + | |FastDigest green buffer 10X | ||
| + | |5μl | ||
| + | |- | ||
| + | |PacI | ||
| + | |2µl | ||
| + | |- | ||
| + | |H<sub>2</sub>O | ||
| + | |2µl | ||
| + | |} | ||
====pPSI dephosphorylation==== | ====pPSI dephosphorylation==== | ||
| + | |||
| + | [[File:Paris_Saclay_140822.jpg|left]] | ||
After 2 hours, add 1.5µL of Alkaline Phosphatase (AP) and let it 1 hour. | After 2 hours, add 1.5µL of Alkaline Phosphatase (AP) and let it 1 hour. | ||
| Line 18: | Line 32: | ||
Then, inactivation of AP at 65°C during 15 minutes. | Then, inactivation of AP at 65°C during 15 minutes. | ||
| - | We've made a electrophoresis to | + | We've made a electrophoresis to check the digestion. |
| - | ( | + | |
| + | '''Legend''' | ||
| + | #pPSI+Apra digested via PacI, dephosphorylated 5µl | ||
| + | #pPSI+Apra digested via PacI, dephosphorylated 5µl | ||
| + | #ladder 5µl | ||
| + | |||
| + | ==== Ligation of BBa_K517003, BBa_K762100, and Geraniol synthase in pPSI==== | ||
| + | |||
| + | {| class="wikitable centre" width="25%" | ||
| + | |+ BBa_K517003 | ||
| + | |- | ||
| + | ! scope=col | components | ||
| + | ! scope=col | volumes | ||
| + | |- | ||
| + | |BBa_K517003 | ||
| + | |10μl | ||
| + | |- | ||
| + | |pPSI | ||
| + | |2μl | ||
| + | |- | ||
| + | |buffer | ||
| + | |2µl | ||
| + | |- | ||
| + | |H<sub>2</sub>O | ||
| + | |5µl | ||
| + | |- | ||
| + | |ligase | ||
| + | |1µl | ||
| + | |} | ||
| + | |||
| + | {| class="wikitable centre" width="25%" | ||
| + | |+ BBa_K5762100 | ||
| + | |- | ||
| + | ! scope=col | components | ||
| + | ! scope=col | volumes | ||
| + | |- | ||
| + | |BBa_K762100 | ||
| + | |10μl | ||
| + | |- | ||
| + | |pPSI | ||
| + | |2μl | ||
| + | |- | ||
| + | |buffer | ||
| + | |2µl | ||
| + | |- | ||
| + | |H<sub>2</sub>O | ||
| + | |5µl | ||
| + | |- | ||
| + | |ligase | ||
| + | |1µl | ||
| + | |} | ||
| + | |||
| + | {| class="wikitable centre" width="25%" | ||
| + | |+ Geraniol Synthase (GS) | ||
| + | |- | ||
| + | ! scope=col | components | ||
| + | ! scope=col | volumes | ||
| + | |- | ||
| + | |GS | ||
| + | |10μl | ||
| + | |- | ||
| + | |pPSI | ||
| + | |2μl | ||
| + | |- | ||
| + | |buffer | ||
| + | |2µl | ||
| + | |- | ||
| + | |H<sub>2</sub>O | ||
| + | |5µl | ||
| + | |- | ||
| + | |ligase | ||
| + | |1µl | ||
| + | |} | ||
| + | |||
| + | And let it in the freezer for 3 hours. | ||
| + | |||
| + | ====Transformation in competent E.coli DH5α==== | ||
| + | |||
| + | After 3 hours, take out the ligation tubes and add 100 µl of competent DH5α in each tube. | ||
| + | |||
| + | Then proceed to the transformation [https://2014.igem.org/Team:Paris_Saclay/Protocols/Transformation_of_competent_e.coli_by_CaCl2 protocol]. | ||
| + | |||
| + | - 20 minutes at 4°C | ||
| + | |||
| + | - 2 minutes 30 seconds at 42°C | ||
| + | |||
| + | - 2 minutes at 4 °C | ||
| + | |||
| + | - Add 0.9 ml of LB into the tubes | ||
| + | |||
| + | Finally put it in a incubator at 37°C for 30 minutes | ||
| + | |||
| + | |||
| + | |||
| + | ====PCR of CAD ==== | ||
| + | With or synthetic gene, we decided to do a PCR to have more matrice. | ||
| + | |||
| + | {| class="wikitable centre" width="25%" | ||
| + | |+ In one tube | ||
| + | |- | ||
| + | ! scope=col | components | ||
| + | ! scope=col | volumes | ||
| + | |- | ||
| + | |H<sub>2</sub>O | ||
| + | |27μl | ||
| + | |- | ||
| + | |5x phusion buffer | ||
| + | |2µl | ||
| + | |- | ||
| + | |dNTP 10 mM | ||
| + | |2µl | ||
| + | |- | ||
| + | |iPS83 10µM | ||
| + | |5µl | ||
| + | |- | ||
| + | |iPS84 10µM | ||
| + | |5µl | ||
| + | |- | ||
| + | |DMSO | ||
| + | |1.5µl | ||
| + | |} | ||
| + | |||
| + | ====PCR purification==== | ||
| + | We use the kit PCR clean-up | ||
| + | |||
| + | the elution volume is 20µl | ||
| - | ==== | + | {| class="wikitable centre" width="25%" |
| - | + | |+ kit PCR clean-up | |
| - | + | ! scope=col | DNA Polymerase | |
| - | + | ! scope=col | volume | |
| - | + | |- | |
| - | + | |Phusion | |
| - | + | |0.5µl | |
| + | |- | ||
| + | |} | ||
| - | + | {| class="wikitable centre" width="25%" | |
| + | |+ PCR cycle | ||
| + | |- | ||
| + | ! scope=col | Temperature (°C) | ||
| + | ! scope=col | time | ||
| + | |- | ||
| + | |98 | ||
| + | |30 sec | ||
| + | |- | ||
| + | |98 | ||
| + | |10 sec | ||
| + | |- | ||
| + | |58 | ||
| + | |30 sec | ||
| + | |- | ||
| + | |72 | ||
| + | |45 sec | ||
| + | |- | ||
| + | |72 | ||
| + | |10 min | ||
| + | |- | ||
| + | |} | ||
| + | ==Photo of the Day== | ||
| + | [[File:Paris Saclay 22_august.jpg|600px|center]] | ||
| + | '''Members present:''' | ||
| + | * Instructors and advisors: Alice, Solenne and Sylvie. | ||
| + | * Students: Eugène, Hoang Vu, Laëtitia, Mélanie, Romain, Sean and Terry. | ||
{{Team:Paris_Saclay/notebook_footer}} | {{Team:Paris_Saclay/notebook_footer}} | ||
Latest revision as of 15:10, 14 October 2014
Contents |
Friday 22nd August
Lab work
Lemon Scent
pPSI digestion
| components | volumes |
|---|---|
| pPSI | 40μl |
| FastDigest green buffer 10X | 5μl |
| PacI | 2µl |
| H2O | 2µl |
pPSI dephosphorylation
After 2 hours, add 1.5µL of Alkaline Phosphatase (AP) and let it 1 hour.
Then, inactivation of AP at 65°C during 15 minutes.
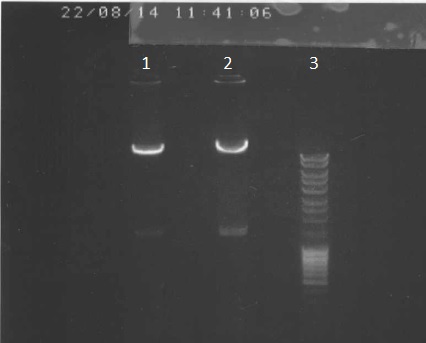
We've made a electrophoresis to check the digestion.
Legend
- pPSI+Apra digested via PacI, dephosphorylated 5µl
- pPSI+Apra digested via PacI, dephosphorylated 5µl
- ladder 5µl
Ligation of BBa_K517003, BBa_K762100, and Geraniol synthase in pPSI
| components | volumes |
|---|---|
| BBa_K517003 | 10μl |
| pPSI | 2μl |
| buffer | 2µl |
| H2O | 5µl |
| ligase | 1µl |
| components | volumes |
|---|---|
| BBa_K762100 | 10μl |
| pPSI | 2μl |
| buffer | 2µl |
| H2O | 5µl |
| ligase | 1µl |
| components | volumes |
|---|---|
| GS | 10μl |
| pPSI | 2μl |
| buffer | 2µl |
| H2O | 5µl |
| ligase | 1µl |
And let it in the freezer for 3 hours.
Transformation in competent E.coli DH5α
After 3 hours, take out the ligation tubes and add 100 µl of competent DH5α in each tube.
Then proceed to the transformation protocol.
- 20 minutes at 4°C
- 2 minutes 30 seconds at 42°C
- 2 minutes at 4 °C
- Add 0.9 ml of LB into the tubes
Finally put it in a incubator at 37°C for 30 minutes
PCR of CAD
With or synthetic gene, we decided to do a PCR to have more matrice.
| components | volumes |
|---|---|
| H2O | 27μl |
| 5x phusion buffer | 2µl |
| dNTP 10 mM | 2µl |
| iPS83 10µM | 5µl |
| iPS84 10µM | 5µl |
| DMSO | 1.5µl |
PCR purification
We use the kit PCR clean-up
the elution volume is 20µl
| DNA Polymerase | volume |
|---|---|
| Phusion | 0.5µl |
| Temperature (°C) | time |
|---|---|
| 98 | 30 sec |
| 98 | 10 sec |
| 58 | 30 sec |
| 72 | 45 sec |
| 72 | 10 min |
Photo of the Day
Members present:
- Instructors and advisors: Alice, Solenne and Sylvie.
- Students: Eugène, Hoang Vu, Laëtitia, Mélanie, Romain, Sean and Terry.
 "
"