Team:Saarland/5 step
From 2014.igem.org
Step 5: Defeat the dark force and bring peace to the galaxy

Skip to a chapter by clicking the corresponding marking above
1. Discussion
Because the high molecular mass hyaluronic acid (HMM-HA) of the naked mole rat seems to be of great importance for treating and preventing cancer, the aim of our project was the recombinant production of this molecule in the novel expression system based on Bacillus megaterium (Tian et al., 2013). One great advantage of the selected bacterium is the already established biosynthetic pathway for the synthesis of HA precursor molecules UDP-D-glucuronic acid and UDP-N-acetyl-D-glucosamine. Consequently the recombinant protein expression of the naked mole rat's hyaluronan synthase 2 (Has2) should be sufficient for HMM-HA production as Has2 catalyses the transformation of UDP-glucuronic acid and UDP-N-acetyl-D-glucosamine to the long, linear hyaluronic acid chain. UDP-glucose 6-dehydrogenases (UDP-GlcDH) converts UDP-glucose to UDP-glucuronate (Garg and Hales, 2004). This reaction represents the limiting step in recombinant HA synthesis in Bacillus subtilis (Widner et al., 2005). Additional expression of UDP-GlcDH should increase the amount of this precursor molecule and push the HMM-HA synthesis towards higher yields.
As our results show, cloning of the two genes has2 and UDP-glcDH into the expression plasmid pSMF2.1 and subsequent transformation into B. megaterium was successful. B. megaterium chassis for the first ever biotechnological production of HMM-HA was established. Nevertheless no HMM-HA production could be detected so far. Neither the secreted form of HMM-HA in the culture medium nor the accumulated form in the cell lysates. Missing HMM-HA production might be due to a misfolded and consequently inactive Has2 or due to inadequate amounts of HMM-HA for detection with the rheometer. A rheometer is a physical measuring instrument to determine the viscosity of solutions (Macosko, 1994). Similar to the human HA, the naked mole rat's HMM-HA is able to bind high amounts of water (Guillaumie et al., 2010). The production of HA should result in an increased viscosity of the medium in case of HA secretion or in an increased viscosity of the cell lysate in case of HA accumulation inside the bacterial cell. Nevertheless viscosity measurements conducted with a rheometer did not show any changes in viscosity. Considering the prior results there might be no hyaluronic acid to be measured. Under the presumption that HA is synthesised the rheometer outcome could have different reasons. The first possibility is that insufficient amounts of HMM-HA are available to be detected by this method. Another aspect is a special property of the hyaluronic acid: the thixotropy. This term means that the longer a fluid undergoes shear stress during rheometer measurements, the lower becomes its viscosity (Hesaraki et al., 2013). This effect opposes the viscosity changes, which are caused among other things mainly by the water binding properties of HA and could have lead to a false negative result. Finally an Enzyme Linked Immunosorbent Assay (ELISA) might be the more sensitive and more reliable detection method for low amounts of HMM-HA and should be preferred in future experiments.
Has2 is a mammalian membrane protein comprising 7 transmembrane domains (Necas, 2008). Its complex structure might be too difficult to be correctly folded and integrated into the bacterial cytoplasm membrane, especially because no chaperons were recombinantly expressed to mediate proper folding. Therefore we suggest the additional cloning of a chaperone coding gene in B. megaterium for future approaches to synthesise HMM-HA. Besides, the usage of Saccharomyces cerevisiae as simple and alternative eukaryotic expression system should be considered, especially in regards to a functional ER and Golgi network. Sec61 mediated cotranslational transport and integration into the ER membrane as well as complex glycosylation processes during protein folding might contribute to the expression of a functional Has2 protein and consequently to HMM-HA production. However, the great drawback of Saccharomyces cerevisiae as expression System is the fact that the yeast cell wall consists of alternative sugar molecules (Ballou, 1982). Precursor molecules for HMM-HA production are not present and a laborious pathway engineering would be necessary to enable the HMM-HA synthesis in Saccharomyces cerevisiae.
Even if there are small amounts of HMM-HA produced under the tested conditions, there are several reasons why HMM-HA could not be detected. B. megaterium is a gram positive bacterium with a complex, high molecular cell wall (Weibull and Bergström, 1958). Therefore the incorporation of Has2 in the membrane and the secretion of the HMM-HA across the cell wall into the culture medium might be difficult. So far two different theories have enunciated how the secretion might work. Either Has2 forms a membrane channel itself or there is an unknown polysaccharide transporter involved.
 Modelling
Modelling
Nevertheless low molecular mass hyaluronic acid (LMM-HA) is recombinantly produced and secreted in high amounts by Bacillus subtilis, a close relative of B. megaterium (Liu et al., 2011; Widner et al., 2005). Fluorescence microscopy with GFP tagged Has2 variant showed dots of higher intensity insight the bacterial cell probably indicating an accumulation of Has2 protein and the HMM-HA itself in insoluble spheric inclusions, also referred as poly hydroxyalkanoate (PHA) inclusion bodies. This accumulation could be cause for the detection problems, too. Results could not be reproduced for specific localisation studies via laser scanning fluorescence microscopy.
A Western Blot was performed with anti-GFP antibodies to confirm the expression of the GFP tagged Has2 protein. As the Western Blot shows, the protein is expressed in very low amounts. Due to the fact that GFP signal was not present anymore even under various expression conditions, it is most likely that Has2 protein is not correctly folded in B. megaterium because of its complex structure. Alternatively the protein could be degraded directly after production.
Aside from UDP-GlcDH a lot of other enzymes are involved in HA precursor biosynthesis (Widner et al., 2005).
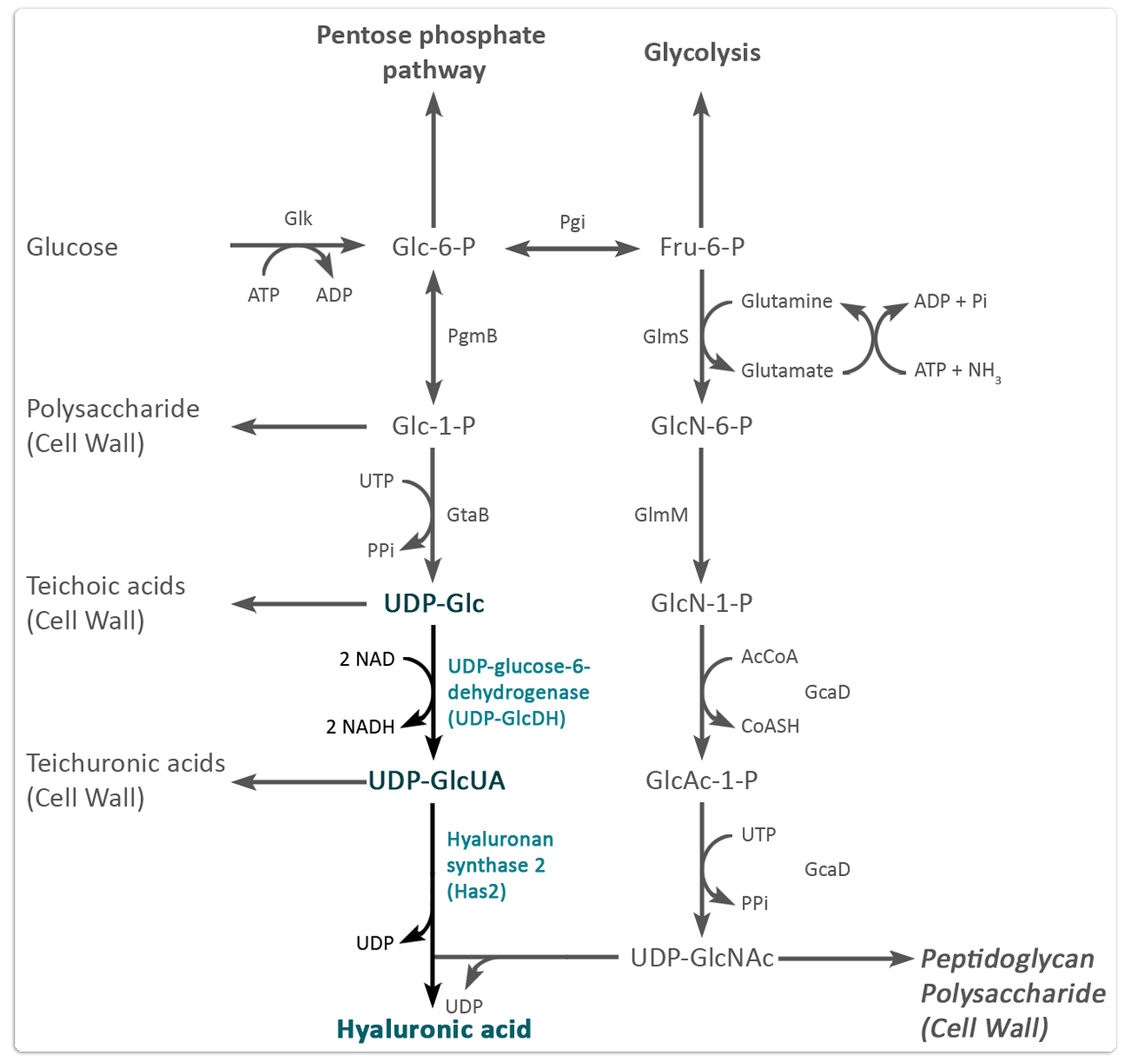
Figure 1 shows the entire biosynthetic pathway for synthesis of HA precursor molecules in B. megaterium. Each of these proteins was amplified from genomic DNA of B. megaterium and is provided by us as BioBricks for future pathway optimisation especially in other expression systems, such as Saccharomyces cerevisiae.
 Biobricks
Biobricks
2. Outlook
The aim of our project was the recombinant production of the naked mole rat high molecular mass hyaluronic acid in B. megaterium. To reach this aim the optimisation of the protein expression and HA production has to be continued. Therefore the proposed genes of the biosynthesis pathway and genes coding for chaperones should additionally be added to the B. megaterium based expression system or other expression systems as described above.
After successful production we further want to purify the HMM-HA to get high amounts of this novel glycosaminoglycane for physiological tests regarding anti-cancer therapy and prophylaxis. Cell culture experiments like proliferation and survival assays will show the therapeutic properties of the naked mole rat HMM-HA, whereas qRT-PCR experiments - including the CD44 receptor pathway for instance - will uncover the mode of action. Alternatively the transfection of human or mouse cell lines with the hyaluronan synthase gene (has2) could be used to confirm the early contact inhibition phenotype and therefore the anti carcinogenic properties of the HMM-HA in human cancer cells. We are confident that the naked mole rat HMM-HA will be applicable in cosmetics and medicine in the near future.
 Modelling
Modelling
 Previous Step
Previous Step
References
Ballou, C. E. (1982). The Molecular Biology of the Yeast Saccharomyces: Metabolism and Gene Expression. CSH Lab Press, Chapter 7 - Yeast Cell Wall and Cell Surface
Garg, H.G., and Hales, C.A. (2004). Chemistry and Biology of Hyaluronan (Elsevier).
Guillaumie, F., Furrer, P., Felt-Baeyens, O., Fuhlendorff, B. L., Nymand, S., Westh, P., Gurny, R., Schwach-Abdellaoui, K. (2010). Comparative studies of various hyaluronic acids produced by microbial fermentation for potential topical ophthalmic applications. J Biomed Mater Res A. 92(4):1421-30.
Hesaraki, S., Borhan, S., Zamanian, A., Hafezi-Ardakani, M. (2013). Rheological Properties and Injectability of β-Tricalcium Phosphate-Hyaluronic acid/Polyethylene Glycol Composites Used for the Treatment of Vesicouretheral Reflux. Advances in Biomedical Engineering Research (ABER) 1(3):40-44
Liu, L., Liu, Y., Li, J., Du, G., and Chen, J. (2011). Microbial production of hyaluronic acid: current state, challenges, and perspectives. Microb. Cell Factories 10, 99.
Macosko, C. W. (1994). Rheology: Principles, Measurements, and Applications. Wiley-VCH.
Necas (2008). Hyaluronic acid (hyaluronan): a review. Vet. Med. (Praha) 53, 397–411.
Tian, X., Azpurua, J., Hine, C., Vaidya, A., Myakishev-Rempel, M., Ablaeva, J., Mao, Z., Nevo, E., Gorbunova, V., and Seluanov, A. (2013). High-molecular-mass hyaluronan mediates the cancer resistance of the naked mole rat. Nature 499, 346–349.
Weibull, C., Bergström, L. (1958) The chemical nature of the cytoplasmic membrane and cell wall of Bacillus megaterium, strain M. Biochimica et Biophysica Acta 30(2), 340-351.
Widner, B., Behr, R., Dollen, S.V., Tang, M., Heu, T., Sloma, A., Sternberg, D., DeAngelis, P.L., Weigel, P.H., and Brown, S. (2005). Hyaluronic Acid Production in Bacillus subtilis. Appl. Environ. Microbiol. 71, 3747–3752.
 "
"



Impressum/Copyright