Team:Aachen/Project/2D Biosensor
From 2014.igem.org
(→Principle of Operation) |
|||
| Line 54: | Line 54: | ||
[[File:Aachen_15-10-14_Principle_of_operation_2D_sensor_ipo.png|right|150px]] | [[File:Aachen_15-10-14_Principle_of_operation_2D_sensor_ipo.png|right|150px]] | ||
| - | =Principle of Operation= | + | == Principle of Operation == |
<span class="anchor" id="biosensorpoo"></span> | <span class="anchor" id="biosensorpoo"></span> | ||
| Line 79: | Line 79: | ||
</center> | </center> | ||
| - | |||
| - | |||
| - | |||
{{Team:Aachen/BlockSeparator}} | {{Team:Aachen/BlockSeparator}} | ||
| Line 101: | Line 98: | ||
[[File:Aachen_14-10-15_Medal_Cellocks_iNB.png|right|150px]] | [[File:Aachen_14-10-15_Medal_Cellocks_iNB.png|right|150px]] | ||
| - | = Achievements = | + | == Achievements == |
<span class="anchor" id="biosensorachievements"></span> | <span class="anchor" id="biosensorachievements"></span> | ||
| - | == Medium == | + | === Medium === |
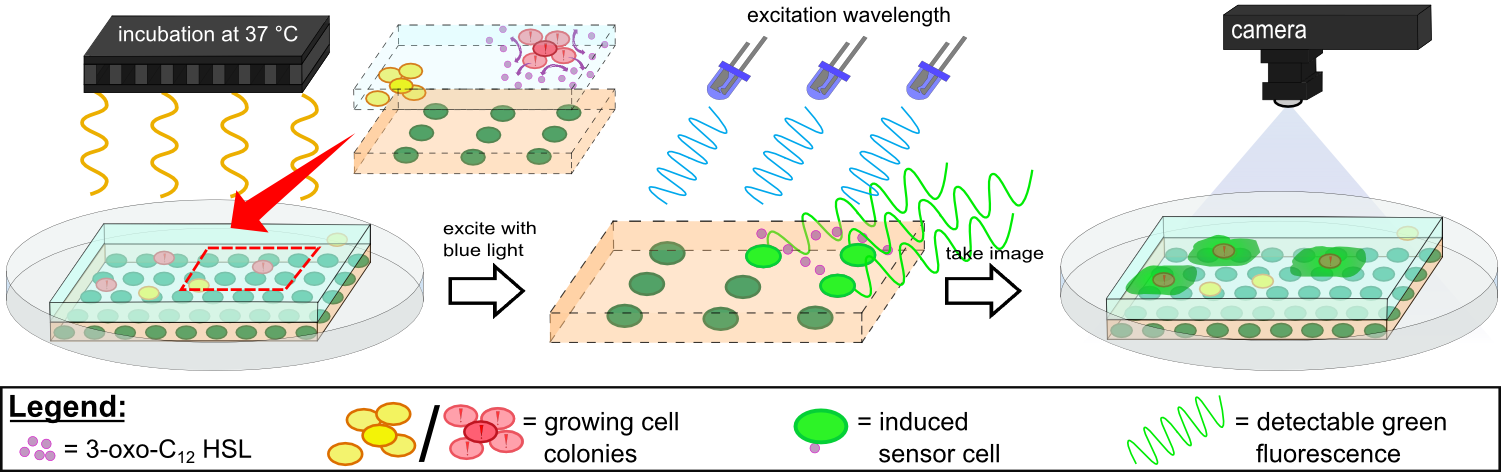
Prior to using our own device for detection of fluorescence emitted by the sensor chips we used equipment readily available in the lab. A Molecular Imager® Gel Doc<sup>TM</sup> XR+ from BIO-RAD was available which used UV and white light illuminators. Only two different filters were availble for the excitation ligth wavelength, which resulted in very limitted possibilities for excitation of fluorescent molecules. For example, it was possible to detect the expression of iLOV in our sensor chips, but in contrast detection of GFP was not possible. Thus the Gel Doc<sup>TM</sup> XR+ was not ideal for our project. | Prior to using our own device for detection of fluorescence emitted by the sensor chips we used equipment readily available in the lab. A Molecular Imager® Gel Doc<sup>TM</sup> XR+ from BIO-RAD was available which used UV and white light illuminators. Only two different filters were availble for the excitation ligth wavelength, which resulted in very limitted possibilities for excitation of fluorescent molecules. For example, it was possible to detect the expression of iLOV in our sensor chips, but in contrast detection of GFP was not possible. Thus the Gel Doc<sup>TM</sup> XR+ was not ideal for our project. | ||
(iLOV_GFP_HM_1,5h.png) | (iLOV_GFP_HM_1,5h.png) | ||
| Line 115: | Line 112: | ||
(5Tage_K131026_neb_tb_1,5h) | (5Tage_K131026_neb_tb_1,5h) | ||
| - | == Agar | + | === Agar Concentration === |
For sensor chip manufacturing an optimal agarose concentration of 1.5% was found. When agarose concentrations below 1.5% (w/v) were used the sensor chips were easily damaged and were not transportable. Agar concentrations above 1.5% (w/v) had to be avoided, because the agarose started to solidify before it could be poured into the chip mold. | For sensor chip manufacturing an optimal agarose concentration of 1.5% was found. When agarose concentrations below 1.5% (w/v) were used the sensor chips were easily damaged and were not transportable. Agar concentrations above 1.5% (w/v) had to be avoided, because the agarose started to solidify before it could be poured into the chip mold. | ||
Agarose was chosen above agar, because of more evely linkage and thus more homogenous chips. In addition agarose reduced diffusion of inducer molecules through the chip. Reduced diffusion was desired in order to achieve distinct fluorescent spots on the sensor chips. | Agarose was chosen above agar, because of more evely linkage and thus more homogenous chips. In addition agarose reduced diffusion of inducer molecules through the chip. Reduced diffusion was desired in order to achieve distinct fluorescent spots on the sensor chips. | ||
| - | == Chip | + | === Chip Form === |
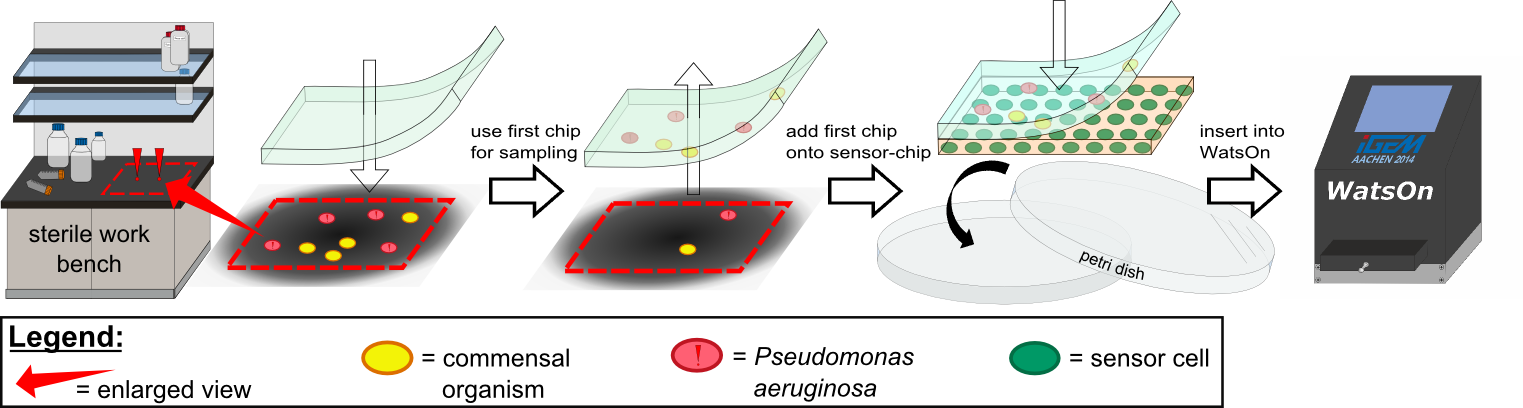
Various approaches were tried for production of sensor chips with reproducable quality. The first approach was to cast | Various approaches were tried for production of sensor chips with reproducable quality. The first approach was to cast | ||
every sensor chip indicidually. In order to archive a plain chip surface, which was required for high quality images, we tried to cast the sensor chips between two microscope slides. This approach had to be rejected, because the agar was too liquid. Second, we produced a closed mold in which the liquid agar was injected using a pipette, but we encountered a high frequency of bubbles in the chips when using this approach. Bubbles in the sensor chips resulted in problems during fluorescence evalutaion. | every sensor chip indicidually. In order to archive a plain chip surface, which was required for high quality images, we tried to cast the sensor chips between two microscope slides. This approach had to be rejected, because the agar was too liquid. Second, we produced a closed mold in which the liquid agar was injected using a pipette, but we encountered a high frequency of bubbles in the chips when using this approach. Bubbles in the sensor chips resulted in problems during fluorescence evalutaion. | ||
| Line 128: | Line 125: | ||
(final_chipform.jpg) | (final_chipform.jpg) | ||
| - | == Induction == | + | === Induction === |
For the induction of the used constructs we use IPTG or 3-oxo-C12 HSL. | For the induction of the used constructs we use IPTG or 3-oxo-C12 HSL. | ||
The sensor cells with K1319042 in BL21 can detect a IPTG concentration of 1 mM (0,2 µl). | The sensor cells with K1319042 in BL21 can detect a IPTG concentration of 1 mM (0,2 µl). | ||
| Line 138: | Line 135: | ||
(Zeitaufnahmen bearbeitet von Arne) | (Zeitaufnahmen bearbeitet von Arne) | ||
| - | ==Testing our | + | === Testing our Sensor Chips with a Platereader === |
{{Team:Aachen/Figure|Aachen_K1319042_Platereader.gif|title=Testing K1319042 in our sensor chips|subtitle=K1319042 in our sensorchip induced with 2 µl iPTG and measured with a Platereader. Blue color indicates no fluorescence, Red color indicates fluorescence.|width=300px}} | {{Team:Aachen/Figure|Aachen_K1319042_Platereader.gif|title=Testing K1319042 in our sensor chips|subtitle=K1319042 in our sensorchip induced with 2 µl iPTG and measured with a Platereader. Blue color indicates no fluorescence, Red color indicates fluorescence.|width=300px}} | ||
{{Team:Aachen/Figure|Aachen_K131026_Platereader.gif|title=Testing K131026 in our sensor chips|subtitle=K131026 in our sensorchip induced with 0.2 µl 3-oxo-C{{sub|12}} HSL and measured with a Platereader. Blue color indicates no fluorescence, Red color indicates fluorescence.|width=300px}} | {{Team:Aachen/Figure|Aachen_K131026_Platereader.gif|title=Testing K131026 in our sensor chips|subtitle=K131026 in our sensorchip induced with 0.2 µl 3-oxo-C{{sub|12}} HSL and measured with a Platereader. Blue color indicates no fluorescence, Red color indicates fluorescence.|width=300px}} | ||
| Line 147: | Line 144: | ||
Detecting Pseudomonas aeruginosa with K131026 in our sensor chip with WatsOn. | Detecting Pseudomonas aeruginosa with K131026 in our sensor chip with WatsOn. | ||
{{Team:Aachen/Figure|Aachen_K131026_Pseudomonas_aeruginosa_detection.gif|title=Detection of ''Pseudomonas aeruginosa'' with K131026|subtitle=Direct detection of ''Pseudomonas aeruginosa on our sensor chips. Sensor cell used were K131026.|width=480px}} | {{Team:Aachen/Figure|Aachen_K131026_Pseudomonas_aeruginosa_detection.gif|title=Detection of ''Pseudomonas aeruginosa'' with K131026|subtitle=Direct detection of ''Pseudomonas aeruginosa on our sensor chips. Sensor cell used were K131026.|width=480px}} | ||
| + | |||
{{Team:Aachen/Footer}} | {{Team:Aachen/Footer}} | ||
Revision as of 07:39, 16 October 2014
|
|
|
 "
"