Development & Optimization
Equipment and medium selection
Our first approach (before developing our own device) was to use the Molecular Imager® Gel Doc™ XR+ from BIO-RAD in our lab to detect fluorescence, which uses UV and white light illuminators. However, only two different filters were available for the excitation light wavelength, which resulted in very limited possibilities for the excitation of fluorescent molecules. For example, it was possible to detect the expression of iLOV in our sensor chips, but in contrast the detection of GFP was not possible. Hence, the Gel Doc™ was not suitable for our project.
We tested different media (i.e. LB, M9, NA, HM and TB) for the preparation of our sensor chips. The details of media composition can be found in the Protocols section. We screened for an optimized media composition that results in minimal background fluorescence and supports cell growth. The resuts of the analysis are presented in the table below. Because of the reduced fluorescence compared to TB medium when using WatsOn for sensor chip evaluation and because of sufficient cultivation conditions for our Cellocks LB medium was chosen over TB medium for sensor hip manufacturing.
| | LB | TB | NA | M9 | HM
|
| Growth of Cellock | + | + | - | - | -
|
| Background fluorescence in GelDoc | + | + | - | - | -
|
| Background fluorescence in WatsOn | - | + | - | - | -
|
Experiments were conducted to test long-time storage of the sensor chips at low temperature and by the addition of glycerol. Storage at -20°C resulted in the loss of our sensor cells. Adding 5-10% (v/v) glycerol ensured survival of the sensor cells, but resulted in an expression stop of fluorescence proteins. Hence, we concluded that long time storage of the sensor chips is not possible under the tested conditions. However, it is possible to store 'ready-to-use' sensor chips for 2 days at 4°C when using LB medium and storage for 5 days was possible with chips made from TB medium.
Optimal agarose concentration for sensor chip manufacturing
For the sensor chip manufacturing, agarose was preferred over agar, because of a uniform linkage that resulted in a better chip homogeneity. In addition, agarose reduced diffusion of the inducer molecules through the chip. A reduced diffusion is vital to observe distinct fluorescent spots on the sensor chips and thus further optimzation of our 2D biosensor was done using agarose-based chips.
Optimal chip configuration
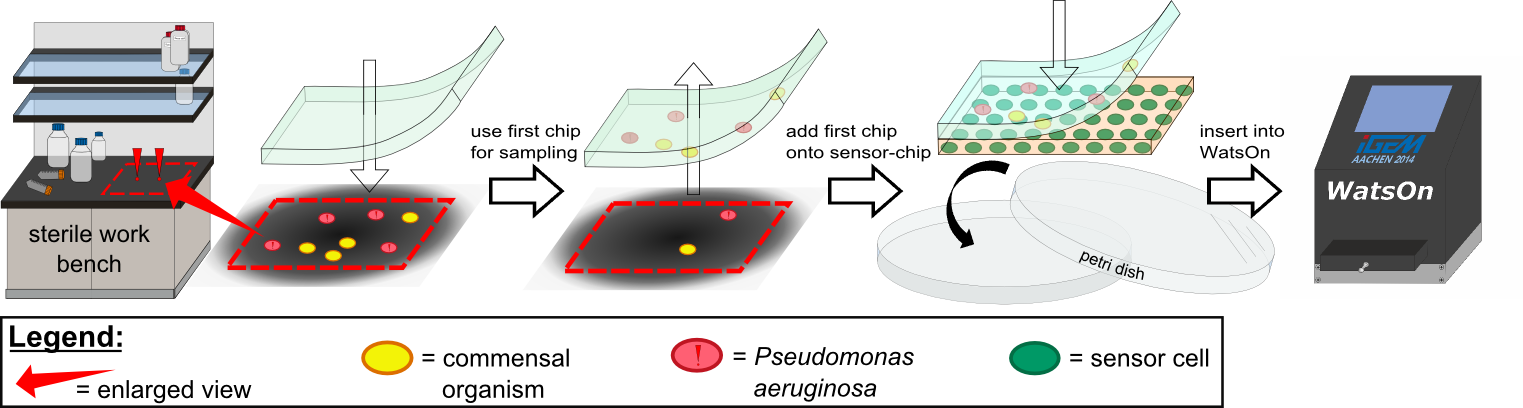
Several approaches were tested for the production of agarose-based sensor chips with reproducible quality. The first approach was to cast every sensor chip individually. To achieve a plain chip surface (a requisite for high quality images), we casted the sensor chips between two microscope slides in an initial attempt. However, this approach was rejected, because the agar was too liquid and leaked from the microscope slides. In the second approach, we prepared a closed mold into which liquid agar was injected with a pipette, but we encountered a high number of bubbles in the resulting chips. Bubbles in the sensor chips interfered with fluorescence evaluation. Finally, we used an open mold into which the agar was poured right after mixing with the sensor cells. Chips were cut out along precast indentations in the casting mold after the agar solidified. An advantage of the open mold was the ability to simultaneously produce nine sensor chips while the surface tension of the liquid agar ensured a plane chip surface.
Induction of the sensor chips
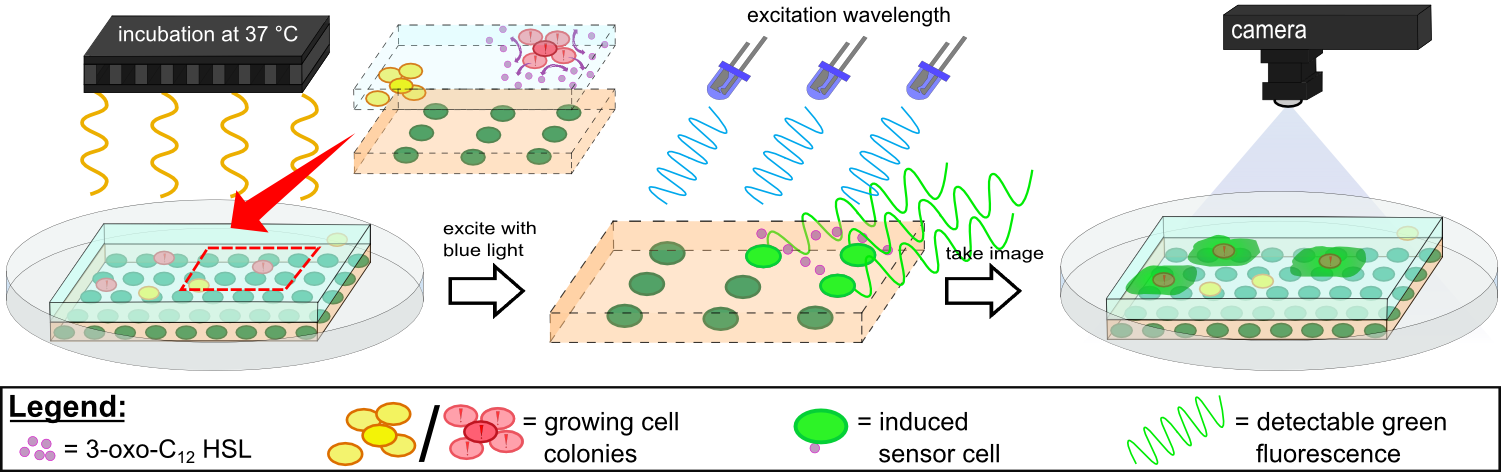
For initial testing of our molecular constructs, we simulated the presence of P. aeruginosa using IPTG or 3-oxo-C12-HSL. However, for induction a minimal volume is required as our initial experiments showed that diffusion of the inducers through the chip hindered the formation of distinct spots on the chips. An optimal and low volume of 0.2 µl was chosen for induction. Sensor cells based on E. coli BL21, which incorporated the K1319042 construct were able to detect IPTG concentrations down to 1 mM (0.2 µl), and as well for the sensor cells based on E. coli BL21 which incorporated the REACh constructs. Sensor cells based on E. coli BL21, which incorporated the [http://parts.igem.org/Part:BBa_K131026 K131026] construct were able to detect HSL concentrations down to 500 µg/ml (0.2 µl). Furthermore, detection of growing P. aeruginosa cells based on secreted HSLs was possible using the [http://parts.igem.org/Part:BBa_K131026 K131026] construct. The experiments conducted during induction of our sensor chips are described in more detail in the Achievements section.
|
 "
"