Team:Aachen/Project/FRET Reporter
From 2014.igem.org
(→Cutting the Fusion Protein with the TEV Protease) |
(→The REACh Construct) |
||
| Line 4: | Line 4: | ||
=The REACh Construct= | =The REACh Construct= | ||
| + | <html><ul class="team-grid"> | ||
| + | <!-- Overview --> | ||
| - | == | + | <li> |
| + | <a class="menulink" href="https://2014.igem.org/Team:Aachen/Project/2D_Biosensor" style="color:black"> | ||
| + | <div class="team-item team-info" ></div> | ||
| + | <div class="team-item team-img" style="background: #102030; norepeat scroll 0% 0% transparent; background-size:100%"> | ||
| + | <div class="menukachel">2D Biosensor</div> | ||
| + | </div> | ||
| + | </a> | ||
| + | </li> | ||
| + | <li> | ||
| + | <a class="menulink" href="https://2014.igem.org/Team:Aachen/Project/FRET_Reporter" style="color:black"> | ||
| + | <div class="team-item team-info" ></div> | ||
| + | <div class="team-item team-img" style="background: #102030; norepeat scroll 0% 0% transparent; background-size:100%"> | ||
| + | <div class="menukachel">REACh Construct</div> | ||
| + | </div> | ||
| + | </a> | ||
| + | </li> | ||
| + | |||
| + | <li> | ||
| + | <a class="menulink" href="https://2014.igem.org/Team:Aachen/Project/Gal3" style="color:black"> | ||
| + | <div class="team-item team-info" ></div> | ||
| + | <div class="team-item team-img" style="background: #0069b8; norepeat scroll 0% 0% transparent; background-size:100%"> | ||
| + | <div class="menukachel">Galectin-3</div> | ||
| + | </div> | ||
| + | </a> | ||
| + | </li> | ||
| + | |||
| + | <li> | ||
| + | <a class="menulink" href="https://2014.igem.org/Team:Aachen/Project/Measurement_Device" style="color:black"> | ||
| + | <div class="team-item team-info" ></div> | ||
| + | <div class="team-item team-img" style="background: #102030; norepeat scroll 0% 0% transparent; background-size:100%"> | ||
| + | <div class="menukachel"><i>WatsOn</i></div> | ||
| + | </div> | ||
| + | </a> | ||
| + | </li> | ||
| + | |||
| + | |||
| + | </ul></html> | ||
| + | |||
| + | == A Fluorescence Answer Faster Than Expression == | ||
| + | <span class="anchor" id="fluorescence"></span> | ||
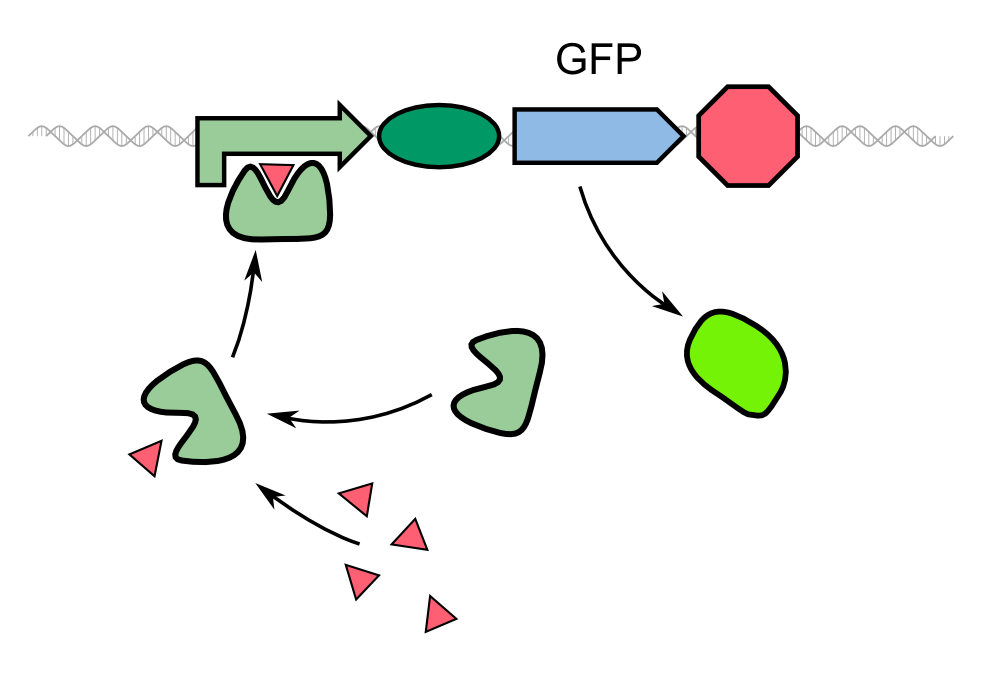
Biosensors often work with a system that is comprised of a reported gene under the control of a promoter that is induced directly by the chemical that the sensor is supposed to detect. In the case of our 2D biosensor for ''Pseudomonas aeruginosa'', the expression of our reporter gene, GFP, would be directly induced by the activity of the bacterium's quorum sensing molecules. However, transcription, translation, folding and post-translational modifications take their time. Since our goal is to detect the pathogen as fast as possible, we wanted to use a system that gives a fluorescent answer fast than just expressing the fluorescent protein. | Biosensors often work with a system that is comprised of a reported gene under the control of a promoter that is induced directly by the chemical that the sensor is supposed to detect. In the case of our 2D biosensor for ''Pseudomonas aeruginosa'', the expression of our reporter gene, GFP, would be directly induced by the activity of the bacterium's quorum sensing molecules. However, transcription, translation, folding and post-translational modifications take their time. Since our goal is to detect the pathogen as fast as possible, we wanted to use a system that gives a fluorescent answer fast than just expressing the fluorescent protein. | ||
| Line 29: | Line 70: | ||
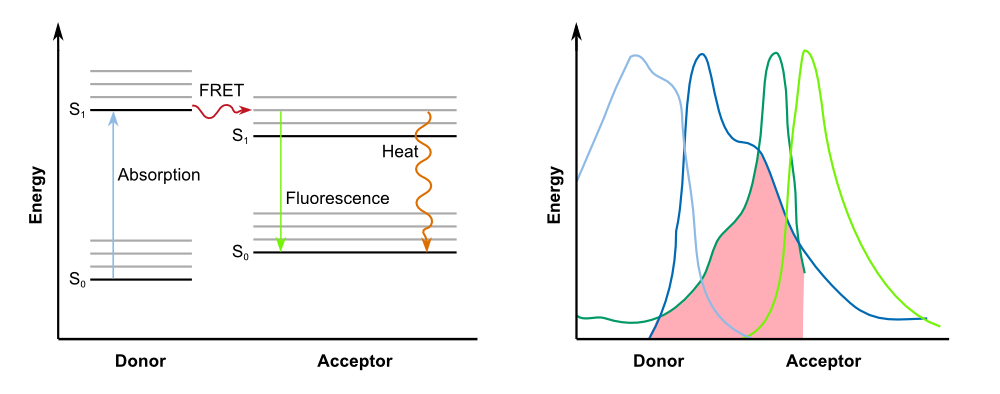
==The FRET (Förster Resonance Energy Transfer) System== | ==The FRET (Förster Resonance Energy Transfer) System== | ||
| + | <span class="anchor" id="fret"></span> | ||
Förster resonance energy transfer (FRET), sometimes also called fluorescence resonance energy transfer, is a physical process of energy transfer. In FRET, the energy of a donor chromophore, whose electrons are in an excited state, is passed to a second chromophore, the acceptor. The '''energy is transferred without radiation''' and is therefore not exchanged via emission and absorption of photons. The acceptor then releases the energy received from the donor, for example, as light of a longer wavelength. | Förster resonance energy transfer (FRET), sometimes also called fluorescence resonance energy transfer, is a physical process of energy transfer. In FRET, the energy of a donor chromophore, whose electrons are in an excited state, is passed to a second chromophore, the acceptor. The '''energy is transferred without radiation''' and is therefore not exchanged via emission and absorption of photons. The acceptor then releases the energy received from the donor, for example, as light of a longer wavelength. | ||
| Line 41: | Line 83: | ||
==REACh Proteins - Dark Quenchers of GFP== | ==REACh Proteins - Dark Quenchers of GFP== | ||
| + | <span class="anchor" id="darkquencher"></span> | ||
In 2006, [http://www.pnas.org/content/103/11/4089.full Ganesan et al.] were the first to present a previously undescribed FRET acceptor, a non-fluorescent yellow fluorescent protein (YFP) mutant called '''REACh (for Resonance Energy-Accepting Chromoprotein)'''. YFP can be used as a FRET acceptor in combination with GFP as the donor in FRET microscopy and miscellaneous assays in molecular biology. The ideal FRET couple should possess a large spectral overlap between donor emission and acceptor absorption - as illustrated in Fig. 2 - but have separated emission spectra to allow their selective imaging. | In 2006, [http://www.pnas.org/content/103/11/4089.full Ganesan et al.] were the first to present a previously undescribed FRET acceptor, a non-fluorescent yellow fluorescent protein (YFP) mutant called '''REACh (for Resonance Energy-Accepting Chromoprotein)'''. YFP can be used as a FRET acceptor in combination with GFP as the donor in FRET microscopy and miscellaneous assays in molecular biology. The ideal FRET couple should possess a large spectral overlap between donor emission and acceptor absorption - as illustrated in Fig. 2 - but have separated emission spectra to allow their selective imaging. | ||
| Line 51: | Line 94: | ||
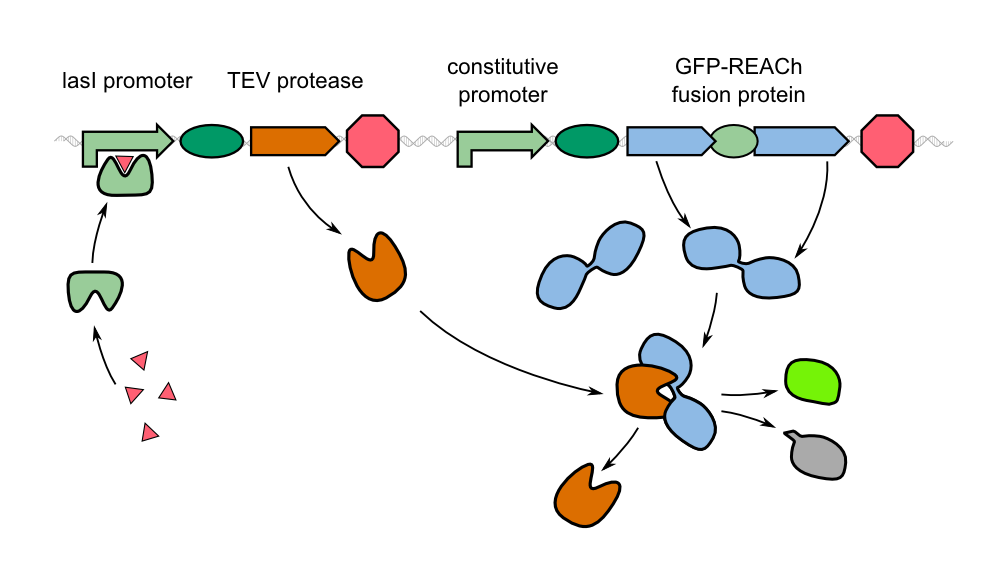
==Producing a GFP-REACh Fusion Protein== | ==Producing a GFP-REACh Fusion Protein== | ||
| + | <span class="anchor" id="gfp-reach"></span> | ||
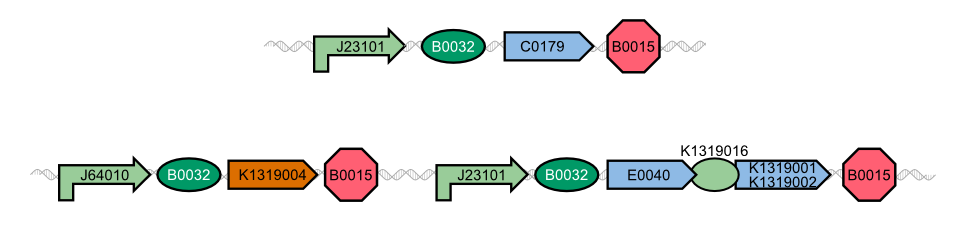
In our project, we reproduced the REACh1 and REACh2 proteins by subjecting an RFC-25 compatible version of the BioBrick [http://parts.igem.org/Part:BBa_E0030 E0030] (EYFP) to a '''QuikChange mutation''', creating the BioBricks K1319001 and K1319002, respectively. Subsequently, we fused each REACh protein with '''GFP (mut3b)''' which is available as BioBrick [http://parts.igem.org/Part:BBa_E0040 E0040]. The protein complex was linked via a '''protease cleavage site''', K1319016. As constitutive promoter we use [http://parts.igem.org/Part:BBa_J23101 J232101]. When GFP is connected to either REACh quencher, GFP will absorb light but the energy will be transferred to REACh via FRET and then emitted in the form of heat; the fluorescence is quenched. Our cells also constitutively express the '''[http://parts.igem.org/Part:BBa_C0179 LasR] activator'''. Together with the HSL molecules from ''P. aeruginosa'', LasR binds to the '''[http://parts.igem.org/Part:BBa_J64010 LasI] promoter''' that controls the expression of the TEV protease, which we make available as K1319004. When the fusion protein is cleaved by the TEV protease, REACh will be separated from GFP. The latter will then be able to absorb and emit light as usual. | In our project, we reproduced the REACh1 and REACh2 proteins by subjecting an RFC-25 compatible version of the BioBrick [http://parts.igem.org/Part:BBa_E0030 E0030] (EYFP) to a '''QuikChange mutation''', creating the BioBricks K1319001 and K1319002, respectively. Subsequently, we fused each REACh protein with '''GFP (mut3b)''' which is available as BioBrick [http://parts.igem.org/Part:BBa_E0040 E0040]. The protein complex was linked via a '''protease cleavage site''', K1319016. As constitutive promoter we use [http://parts.igem.org/Part:BBa_J23101 J232101]. When GFP is connected to either REACh quencher, GFP will absorb light but the energy will be transferred to REACh via FRET and then emitted in the form of heat; the fluorescence is quenched. Our cells also constitutively express the '''[http://parts.igem.org/Part:BBa_C0179 LasR] activator'''. Together with the HSL molecules from ''P. aeruginosa'', LasR binds to the '''[http://parts.igem.org/Part:BBa_J64010 LasI] promoter''' that controls the expression of the TEV protease, which we make available as K1319004. When the fusion protein is cleaved by the TEV protease, REACh will be separated from GFP. The latter will then be able to absorb and emit light as usual. | ||
| Line 60: | Line 104: | ||
==Cutting the Fusion Protein with the TEV Protease== | ==Cutting the Fusion Protein with the TEV Protease== | ||
| + | <span class="anchor" id="TEVprotease"></span> | ||
To cut the GFP_REACh fusion protein off, we chose '''Tobacco Etch Virus (TEV) protease''', a highly sequence-specific cysteine protease, that is frequently used for the controlled cleavage of fusion proteins ''in vitro'' and ''in vivo''. The native protease also contains an internal self-cleavage site. This site is slowly cleaved to inactivate the enzyme. The physiological reason for the self-cleavage is unknown, however, undesired for our use. Therefore, our team uses a variant of the native TEV protease that contains the mutation S219V which results in an alteration of the cleavage site so that self-inactivation is diminished. | To cut the GFP_REACh fusion protein off, we chose '''Tobacco Etch Virus (TEV) protease''', a highly sequence-specific cysteine protease, that is frequently used for the controlled cleavage of fusion proteins ''in vitro'' and ''in vivo''. The native protease also contains an internal self-cleavage site. This site is slowly cleaved to inactivate the enzyme. The physiological reason for the self-cleavage is unknown, however, undesired for our use. Therefore, our team uses a variant of the native TEV protease that contains the mutation S219V which results in an alteration of the cleavage site so that self-inactivation is diminished. | ||
Revision as of 06:56, 13 October 2014
|
|
|
|
|
 "
"