Team:Aachen/Interlab Study
From 2014.igem.org
(→Interlab Study) |
|||
| Line 4: | Line 4: | ||
==Experimental Design== | ==Experimental Design== | ||
| + | For the interlab study, we are testing GFP-containing BioBricks for fluorescence and optical density. As BioBricks we chose I20260, J23101.E0240 and J23115.E0240. The latter consists of a [http://parts.igem.org/Part:pSB3K3 pSB3K3] backbone with an insert, a combination of the promoter [http://parts.igem.org/Part:BBa_J23101 J23101], the RBS [http://parts.igem.org/Part:BBa_B0032 B0032], the GFP coding sequence [http://parts.igem.org/Part:BBa_E0040 E0040] and the terminator [http://parts.igem.org/Part:BBa_B0015 B0015]. J23101.E0240 has the same insert as I20260, but has [http://parts.igem.org/Part:pSB1C3 pSB1C3] as a backbone. J23115.E0240 only differs from J23101.E0240 in the use of another promotor, namely [http://parts.igem.org/Part:BBa_J23115 J23115]. As a negative control, we used just B0015 in pSB1C3. | ||
| + | |||
| + | Over a time span of 18 hours the optical density and fluorescence of cultures containing these BioBricks were measured every 2 hours using the spectrophotometer and plate reader, respectively. | ||
| + | |||
| - | |||
==Expectation== | ==Expectation== | ||
| - | J23101.E0240 and | + | Fluorescence was expected to develop in cultures containing I20260, J23101.E0240 and J23115.E0240, as all include the GFP coding sequence. However, the signal was expected to be stronger in J23101.E0240 than in I20260 since pS1C3 is a high copy plasmid while pSB3K3 is a low to mid copy plasmid. Therefore, a higher fluorescence was expected of J23101.E0240 compared to I20260 even though they share the same insert. J23115.E0240, too, was supposed to produce a fluorescent signal, but J23115 (the mutated version K823012 was used) is a lot weaker promotor than J23101. Therefore, a lot lower fluorescence is expected with this BioBrick, if any. |
| - | + | B0015 was used as our negative control as the insert only contains a terminator and no expression cassette for GFPmut3b, and therefore no fluorescence was expected. | |
| - | |||
==Results== | ==Results== | ||
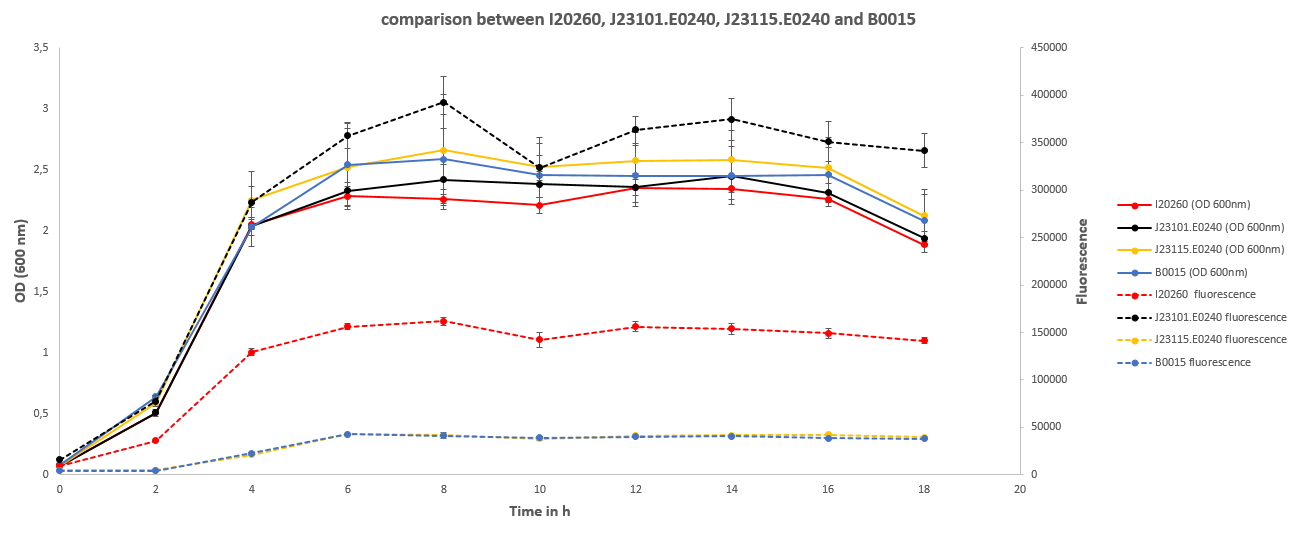
| - | After observing the | + | After observing the optical density (OD) and fluorescence for 18 hours while taking samples every 2 hours, the following results were obtained: |
[[File:Interlabstudy_overview_wiki.png|1000px]] | [[File:Interlabstudy_overview_wiki.png|1000px]] | ||
| - | This shows that all cultures had the same | + | This shows that all cultures had the same ODs throughout the experiment. After the exponential growth phase the stationary phase started shortly after 4 hours of cultivation time. The OD did not change from thereon until a cultivation time of 16 hours after which it started to decline. |
| - | The fluorescence followed largely the pattern of the OD, but differed a lot in between the different cultures. J23101.E0240 | + | The development of fluorescence followed largely the pattern of the OD, but differed a lot in between the different cultures. J23101.E0240 exhibited fluorescence three times stronger than I20260, and about 10 times stronger than B0015 and J23115.E0240. The latter two did not differ in terms of fluorescent signal. |
==Discussion== | ==Discussion== | ||
| - | The OD is an indirect measurement of the biomass in the shake flask. Through the correlation of both measurements the results show that the biomass of | + | The OD is an indirect measurement of the biomass in the shake flask. Through the correlation of both measurements the results show that the difference in biomass of the cultures is not significantly enough to affect the fluorescence data. Therefore, the fluorescence data can be interpreted as a direct result of the fluorescence per cell instead of an overall fluorescence per culture. |
| - | + | ||
| - | + | ||
| - | + | The fluorescence data shows a strong difference between the I20260 and J23110.E240. Even though both inserts are the same, there is a difference in fluorescence, as expected, because of the different plasmid backbones. The high copy plasmid pSB1C3 shows a 3 times stronger fluorescence signal per cell than the low to mid copy plasmid pSB3K3. This can be directly related to the number of plasmids in the cells coding for GFP. | |
| - | J23115.E0240 in its original, non mutated state was supposed to show a slight | + | Both J23115.E0240 and B0015 show no significant fluorescence. The increase at 4 hours is explained by the increase of OD resulting in noise. B0015 behaves therefore as expected. J23115.E0240 in its original, non-mutated state was supposed to show a slight but weaker fluorescence than J23101.E0240. However, the mutations introduced made the promoter non-functional, which lead to no expression of GFP and therefore no observation of fluorescence. |
==Materials and Methods== | ==Materials and Methods== | ||
Revision as of 13:07, 3 October 2014
|
 "
"