Team:Freiburg/Content/Results/Receptor
From 2014.igem.org
The Receptor
The specificity of our system is based on the murine CAT-1 receptor, which serves as the viral entry site of our vector into target cells. We could show that our viral vector system can be used to stably integrate genes into the genome of murine cells. We could also target any other cell line by transferring the gene for the murine CAT-1 receptor in these cells before transduction. Since mCAT-1 is naturally only present in murine cell lines, we use human cell lines for transfection of the receptor gene. Expression of a reporter demonstrates that only the cells expressing the mCAT-1 receptor are infected by the viral particles, thus making particular gene transfer possible.
Localisation Receptor
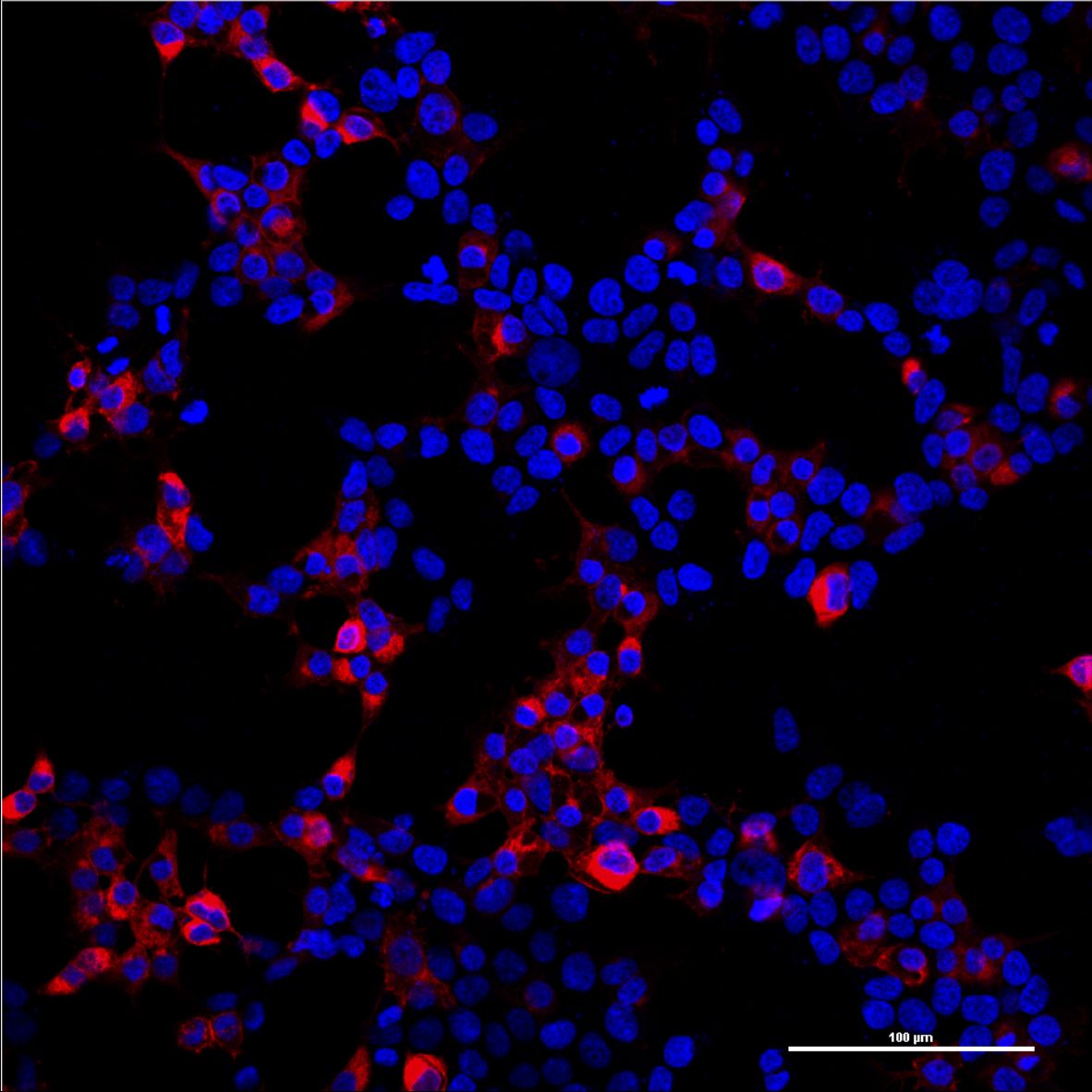
Since the mCAT-1 receptor serves as the entry site of our viral vector, it is essential that it is expressed on the surface of target cells. In order to determinate the localization of the mCAT-1 receptor, we labeled the C-terminus of the mCAT-1 with the fluorescence protein mCherry and transfected this construct in human embryonic kidney (HEK-293T) cells. After distinct time points cells were imaged with a confocal scanning laser microscope. The mCAT1-mCherry was found predominantly at the surface of the cells (Fig. 1-2).
Receptor Functionality
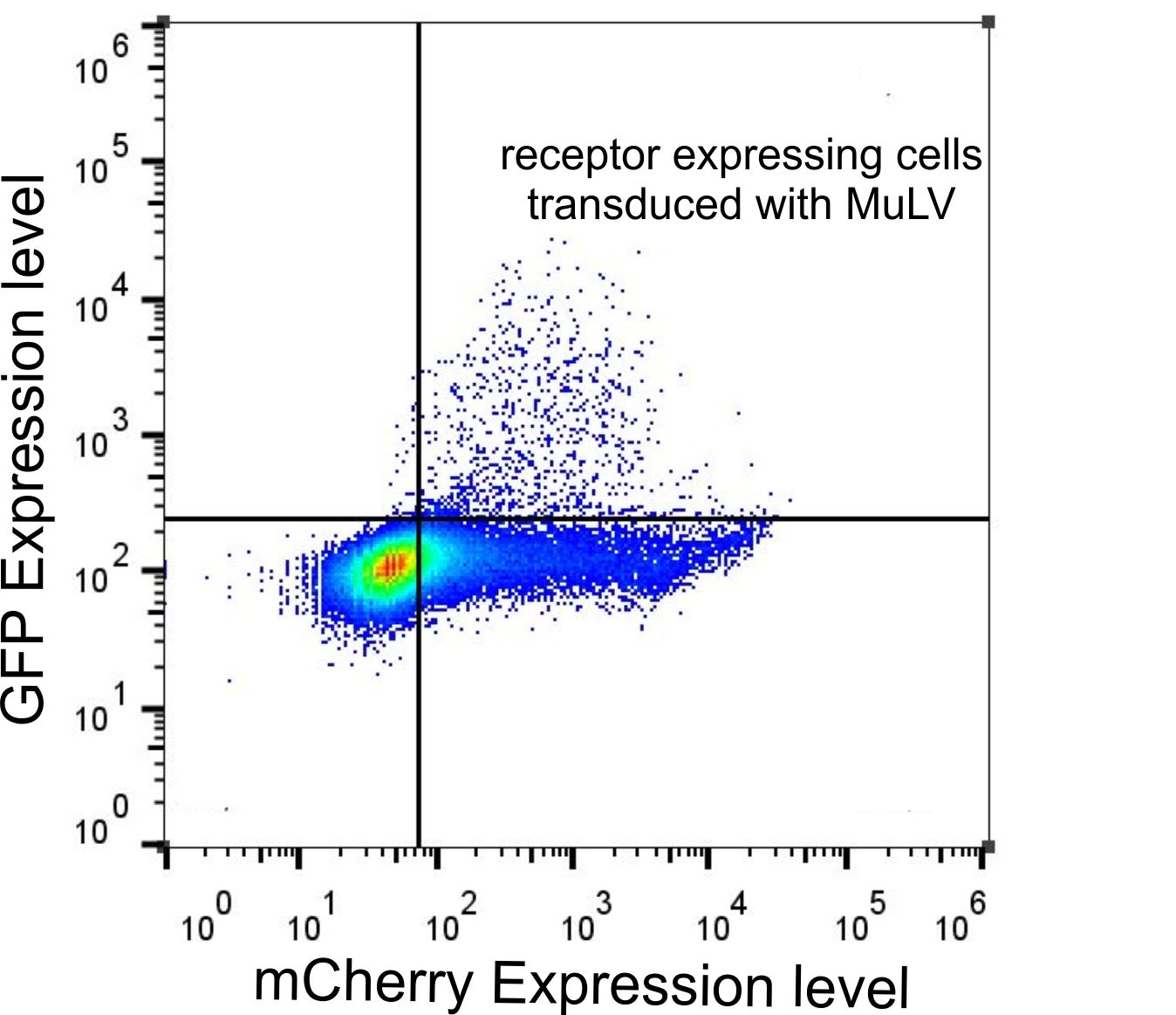
Transduction of genes into murine cell lines which naturally express the mCAT-1 receptor occurred with an efficiency over 80%. For our system we need a functional expression of the receptor in non-murine cell lines, i.e. the receptor has to serve as an entry site for the virus, and the virus has to efficiently deliver its cargo into the target cell. We tested the functionality of the mCAT-1 receptor by infecting HEK-293T cells expressing the receptor with the virus containing eGFP as a cargo. The presence of green fluorescent cells in the infected cultures of different non-murine cell lines indicates that the receptor is not only expressed but can be also used as an entry site by the virus.
During our projekt we generated different receptor constructs all of them labeled with different markers. Since we didn't know, if these tags have an influence on viral infection capabilities, we tested all of them for their functionality determining that all receptor constructs lead to viral infection if expressed in non murine cell lines (Fig.4-5). For making the receptor visible for fluorescent microscopy and analysis by flow cytometry, we labelled the receptor with mCherry (p14rz_005 and p14rz_006). Analysis of cells expressing mCAT-1-mCherry that were infected with MuLV EGFP showed that all cells which were transduced by the viral particles also express the receptor, i.e. only cells expressing the receptor were infected (Fig. 3).

Figure 4: HEK-293T cells transfected with different receptor constructs and infected with MuLV eGFP afterwards.
The cells were transfected with pQCXIN (original vector containing SLC7A1) (A), p14rz_004 (labeled with HA-tag)(B) and p14rz_006 (labeled with HA-tag and mCherry)(C). Cells were transduced with MuLV after 24 h. Pictures were taken after 48 h.

Figure 5: HEK293T cells expressing mCAT-1-mCherry transduced with MuLV-GFP.
Overlay of all three channels (A); DAPI stained nuclei (B); EGFP expression in infected cells (C); mCAT-1-mCherry expression on the surface of HEK293T cells (D) Objective plan apo 60x, 1.4 NA.
Regarding the expression rate of the receptor on the surface of HEK293T cells, we noticed that mCAT-1-mCherry is less present after transduction with our viral particles. That means that the receptor is eather internalized after infection by a mechanism initiated by the virus, or the expression rate of the receptor decreases after infection. Since the particles can only transfer their cargo into dividing cells we found often pairs of cells that were infected by the viral vector or at least high expression rate of the receptor in cells next to targeted cells.
Receptor Expression
The time point for viral infection was adjusted to the time the receptor needs for expression in target cells. In order to determine the expression time of mCAT-1 in HEK-293T cells, we transfected the cells with the HA-tagged mCAT-1 (p14rz_006). Cells transfected with receptor DNA were analyzed after different incubation times. As the expression of the receptor peaks at 24 h after transfection, we used this time point for viral infections (Fig. 6). Since the presence of mCAT-1 on the cell surface is a main factor for viral tranduction efficiency we tested the expression rate of the receptor after transfection of HEK293T cells with different receptor DNA concentrations with Western blot. Although we determined that the presence of the receptor in the cell increases, if more DNA was transfected into the cells, we did not uses such high DNA concentrations. We found that non-murine cells transfected with high amoounts of receptor DNA die due to overexpression of mCAT-1.

Figure 6: Expression time of the receptor that was transfected into HEK293 cells.
After transfection with p14rz_006 (HA-labeled) cells were lysed with RIPA buffer at distinct time points. A Western blot was performed using anti-HA antibody.

Figure 7: Tranfection of HEK-293T cells with different receptor DNA concentrations.
Cells (on 35mm plates) were transfected with 0,6 µg to 5,4 µg p14rz_006 (HA-labeled) per well. Cells were lysed with RIPA buffer after 24 h of incubation and analysed by Western blotting.
 "
"