Team:Heidelberg/pages/PCR 2.0
From 2014.igem.org
Contents |
Introduction
The invention of the polymerase chain reaction in 1983 by Kary Mulliy revolutionized the modern world by enabling amplification of DNA in an exponential manner. Further improvements including the use of thermo-stable DNA-polymerases made the method even more efficient, allowing its widespread use in nearly every field of modern diagnostics and research. However a major part of information is lost when using conventional PCR due to missing transfer of DNA modifications. These modifications are known to be present in bacteria as well as eukaryotes, where they influence the phenotypic expression/transcriptional state of genes. Hence, they represent an additional level of complexity, which is studied in the field of Epigenetics.
In mammals the methylation of cytosine nucleotides that are followed by guanines (CpG) is a prominent key regulator of transcription, embryonic development, X chromosome inactivation and many other cellular functions. (Robertson, K. D. DNA methylation and human disease. Nat. Rev. Genet. 6, 597–610 (2005)). Due to its great importance, the resulting 5-methylcytosine (5mC) is also known as the “fifth base” of eukaryotic genomes. (Lister, R. & Ecker, J. Finding the fifth base: genome-wide sequencing of cytosine methylation. Genome Res. 959–966 (2009)) Disregulations of Cytosine methylation have been reported to play a crucial role in the development of numerous diseases, including cancer, imprinting diseases and repeat-instability diseases including morbus huntington (Robertson, K. D. DNA methylation and human disease. Nat. Rev. Genet. 6, 597–610 (2005))
Even though the detection and mapping of methylation patterns has become feasible in a high-throughput manner by using bisulfite sequencing and array techniques (Meissner, A. et al. Genome-scale DNA methylation maps of pluripotent and differentiated cells. Nature 454, 766–70 (2008)), further functional analysis is often limited due to the small amount of primary material or would imply de-novo synthesis of characterized templates.
We propose the PCR 2.0 as an easy and efficient way to amplify DNA templates in an exponential manner while maintaining their specific methylation pattern. The pivotal element of this PCR is the use of a heat stable DNA methyltransferase (DNMT), which preserve the methylation pattern of the parental strand during replication. Although DNA methyltransferases exist in thermophile organisms (Watanabe, M., Yuzawa, H., Handa, N., & Kobayashi, I. (2006). Hyperthermophilic DNA methyltransferase M.PabI from the archaeon Pyrococcus abyssi. Applied and Environmental Microbiology, 72(8), 5367–75.) until now no suitable protein could be found or synthesized that withstands the harsh conditions of a PCR.
The main goal of our project has therefore been the generation of a heat stable DNMT that can be produced in a large scale and shows efficient and specific methylation of DNA. Our approach applies protein circularization by using self-splicing protein sequences, socalled inteins from our toolbox as well as our linker design software (links). Since the restriction of conformational changes through intramolecular bonds has been known to increase the stability of proteins (Vieille, C. & Zeikus, G. Hyperthermophilic enzymes: sources, uses, and molecular mechanisms for thermostability. Microbiol. Mol. Biol. Rev. 65, 1–43 (2001)) and joining of C and N terminus were reported to increases in thermostability of smaller peptides (references xylanase gfp…) we started the circularization of the so far biggest protein…
Background
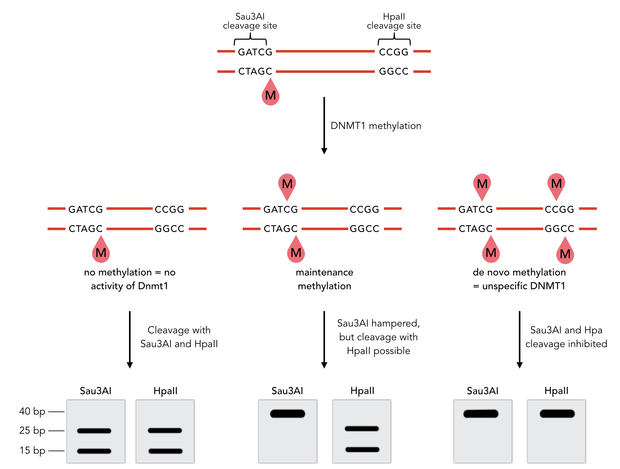
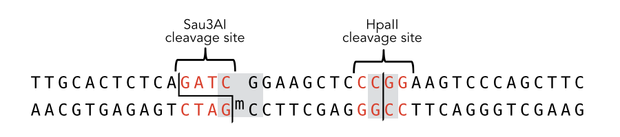
In mammals, DNA methylation plays an active role in gene regulating and cell memory. It mainly occurs at CpG dinucleotides in promoter regions or in repetitive sequences of the DNA where a methyl group (CH3) is covalently linked to the 5’ end cytosine on both complementary DNA strands. Depending on the numbers and positions of the methyl groups, gene expression can be enhanced or silenced, giving each cell an individual protein expression pattern and subsequent function. A family of enzymes called DNA methyltransferases (DNMTs) are responsible for the establishment and maintenance of cell-type specific DNA methylation patterns. The enzymes DNMT3a and DNMT3b have a role in de novo methylation of DNA during development, whereas DNMT1 preserves this methylation pattern in every cell cycle by transferring the methylation pattern from the parental DNA strand to the new daughter strand. (1)
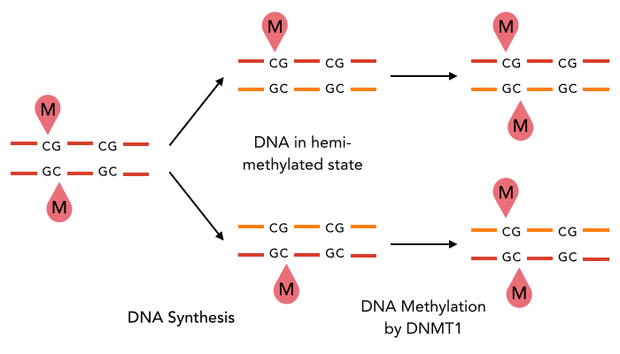
After semiconservative replication, only one DNA strand contains the epigenetic information. DNMT1 recognises hemi-methylated DNA and transfers a methyl group from the donor molecule S-adenosyl-L-methionine (SAM) to the C5 position of the cytosine residue on the complement DNA strand, creating a 5-methylcytosine.
DNA methylation is a precondition for cell differentiation and a deviation in the pattern may lead to malfunction of the cell, resulting in cancer growth, mental illnesses and other diseases (6). Large efforts were made to decipher human DNA methylation patterns. Although now it can be detected (reviewed in (7)), direct amplification of methylated DNA fragments, e.g. for functional analysis in vitro, is impossible up to now. Our project aims facilitate epigenetic research by developing a more stable DNMT1 enabling amplification of methylated DNA.
Circularisation of Dnmt1
Dnmt1 is of particular interest for a methylation maintenance PCR, as it allows exact duplication of the original methylation pattern to every new DNA copy without introducing de novo methylation.
PCR uses three different temperatures for denaturing, annealing and elongation of the DNA sequence, ranging from 55°C to 98°C, requiring extremely heat enzymes.
Circularisation is one way to increase heat stability of a protein, because it anchors the loose terminal ends to the rest of the protein This decreases the high mobility of the regions and therefore making them less affected for denaturing. Further information can be found at the circularisation toolbox element.
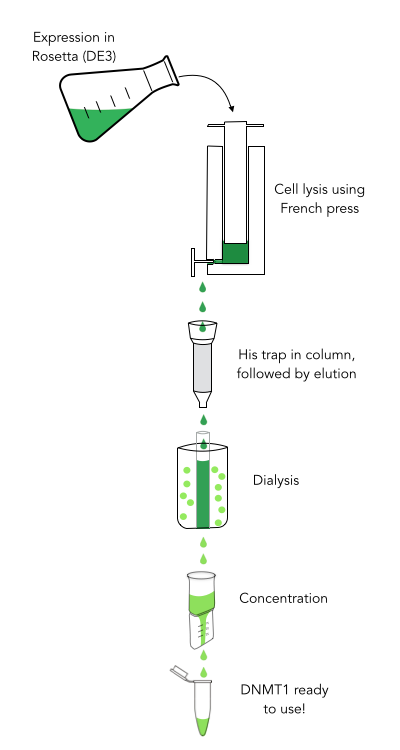
For our experiments we used a truncated version of DNA methyltransferase (cytosine-5) 1 of Mus musculus (713-1602 amino acids) kindly provided by Dr. Pavel Bashtrykov. In contrast to the full-length Dnmt1 that can only be expressed in a baculovirus-insect cell system, this truncated Dnmt1 can also be expressed in E.coli. Moreover, the N and C terminal ends of Dnmt1 (731-1602) are closer together than in the full-lengh Dnmt1. This likely facilitates circularization and might cause less deformation and a lower impact on overall activity. Dnmt1(731-1602) contains the C-terminal catalytic domain and the aligning bromo-adjacent homology (BAH) domains, which prevents the binding of unmethylated DNA in the catalytic core. The enzyme is missing the CXXC domain, which has a high affinity to CpG hemi-methylated dinucleotides, but was shown to be dispensable for protein function (2). The truncated version has almost the same methylation activity and specificity as the wild type, as shown by our own experiments and by Bashtrykov and colleagues (2).
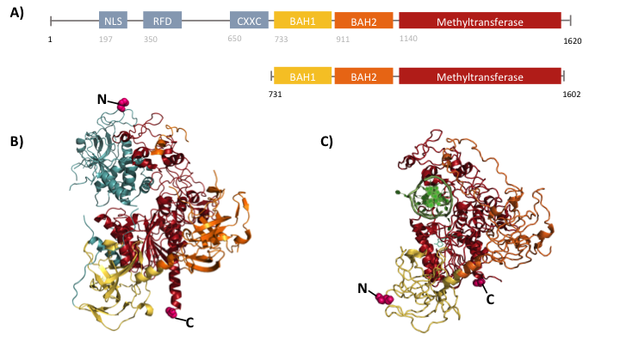
Structural overview of full length mDNMT1 and truncated mDNMT1(650–1602) (A) Color-coded domain devision and numbering of mDNMT1 sequence. (B) Ribbon presentation of full-length DNMT1 with the CXXC,BAH1, BAH2,and methyltransferase domain colored in light blue, yellow, orange and red, respectively. (C) Crystal structure of truncated DNMT1, missing the CXXC domain. Distant between terminal ends of the proteins are closer than at the full length version.
However, even the truncated Dnmt1 is a very large protein of 100kDa and consists of 872 amino. A protein this size has never been circularised before. According to the crystal structure, the distance between the termini of the truncated Dnmt1 is 48 Å.
We suspected circularization by direct fusion of both termini might cause a strong deformation of the Dnmt1 structure leading to a loss of function. Therefore we used a linker amino acid sequence, adapted to the structure of Dnmt1: it needed to be long enough to bridge the gap of 48 Å and bypassing the catalytic core located prominently in the middle of the 3 dimensional enzyme structure.
For circularisation we used NpuDnaE Intein, very efficient inteins for splicing the terminal ends together, as we already tested in circularisation of GFP, Lysosyme and Xylanse.
We tried two different linkers: one rather rigid linker, calculated by a previous version of our linker software subsequently optimised through experience, and one flexible linker.
Material and methods
Results
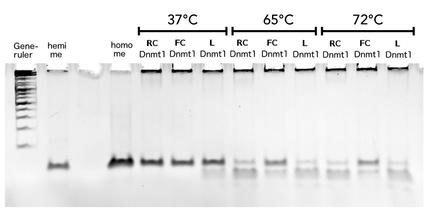
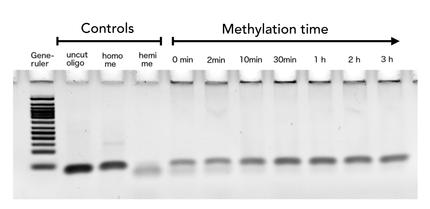
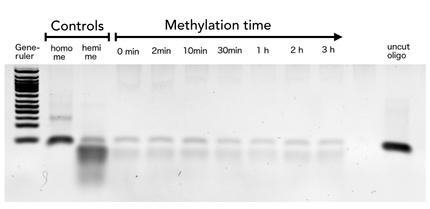
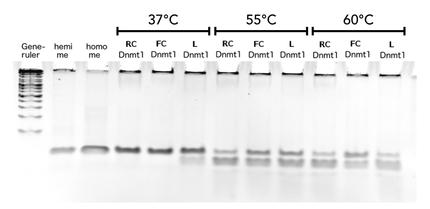
Circular DNMT1 with rigid linker (RC), circular DNMT1 with flexible linker (FC) and linear DNMT1 were heat shocked for 5 sec at 65°C and 72°C.Methylation after one hour was measured. A darker upper band for FC is even visible by eye, indication higher methylation activity.
 "
"