Team:SUSTC-Shenzhen/Project
From 2014.igem.org
Project Description
a small overview to the whole big ideas
Contents |
Introduction
In this project, we intended to establish a synthetic biology-based effective HIV-curing system with less side-effects. The key goal is to integrate CRISPR/Cas system into human blood system to protect CD4+ cells against viral infection. The gRNA is designed to target the relative conserved regions in HIV viral genome and inactivate its biological activity. Since viral vectors seem to be of limited use in gene therapy strategies (e.g., potential pathogenicity), there is in dire need of a simple, efficient system for targeting cell-specific introduction of nucleic acids. By using non-viral DNA delivery system such as A-B-toxin-GAL4 fusion protein, we can deliver plasmids encoding gRNA into the CD4+ cells and readily attack multiple HIV genome sites simultaneously with the most up-to-date knowledge of HIV epidemic.
Background
Written by Yicong Tao, Lin Le & Yongkang Long
HIV (Human immunodeficiency virus)
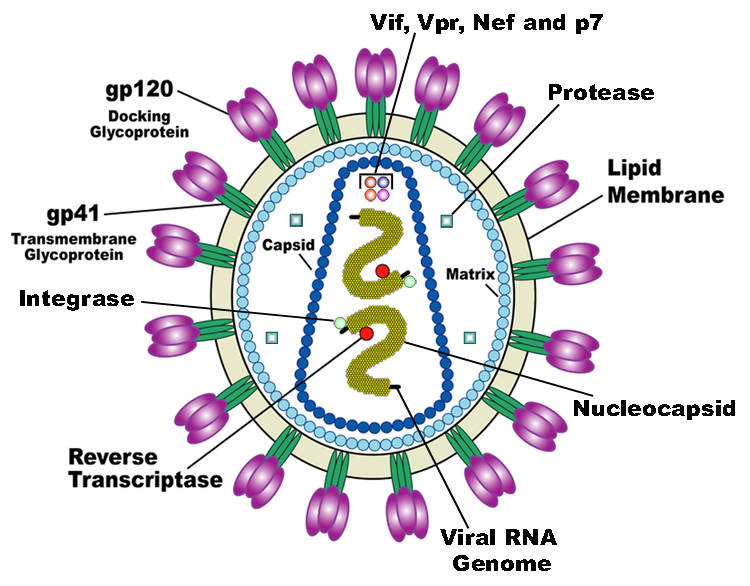
HIV is the trigger of the lethal disease AIDS. It replicates in and kills helper T cells, weakening human immune system and allowing life-threatening opportunistic infection and cancers to thrive. HIV infects vital cells in the human immune system such as helper T cells (especially CD4+ T cells), macrophages, and dendritic cell. As a retrovirus, HIV reverse transcribes its genome and integrated it into the host cell genome after infection. Figure 1 demonstrates the schematic structure of HIV virus (Source: Wikipedia).
Traditional therapies like chemotherapy or radiotherapy can’t protect the helper T cells from virus invasion and the virus can lurk in the helper T cells; drug treatments like HAART (Highly Active Antiretroviral Therapy) can only control the development of the disease and they also have strong side-effects. The process of reverse transcription is extremely error-prone, and the resulting mutations may cause drug resistance or allow the virus to evade the body's immune system.
CRISPR/Cas system
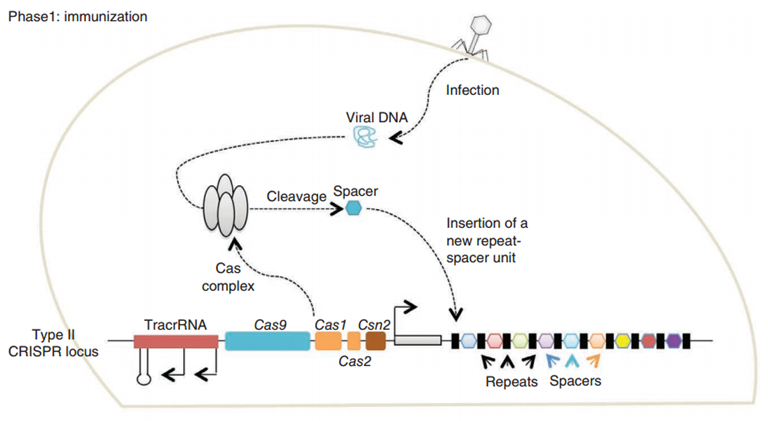
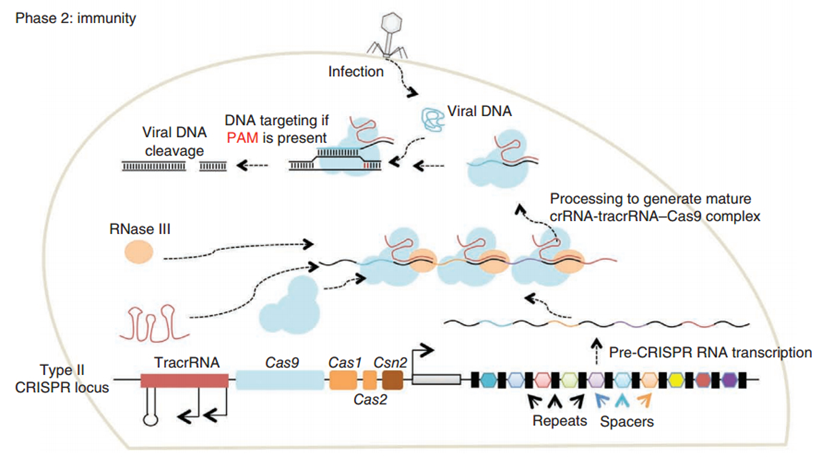
CRISPR/Cas (Clustered regularly interspaced short palindromic repeats/CRISPR-associated system) is originally a self-protecting mechanism in bacteria against external DNA such as virus genome or plasmids. It uses CAS complex to cut the external viral or plasmid DNA and integrating a short DNA segment into the CRISPR loci in the bacteria genome. This short DNA segment will be transcribed into pre-crRNA and bind to another CAS endonuclease called Cas9 with the help of tracrRNA. The Cas9 endonuclease cuts the external DNA in the existence of not only crRNA but also a special recognizing sequence at the 3’ end of the target DNA called PAM (Protospacer Adjacent Motif). When the same external DNA invades next time, the Cas9-tracrRNA-crRNA complex will recognize and cut it in order to destroy its biological activity. Figure 2 demonstrates the mechanism of Type II CRISPR/Cas system in bacteria (Mali, Esvelt, & Church, 2013). Dr. George Church (Mali, Yang, et al., 2013) and Dr. Feng Zhang (Cong et al., 2013) both have successfully transfected Type II CRISPR-Cas system into human cells, which is the foundation of this project.
A-B toxin
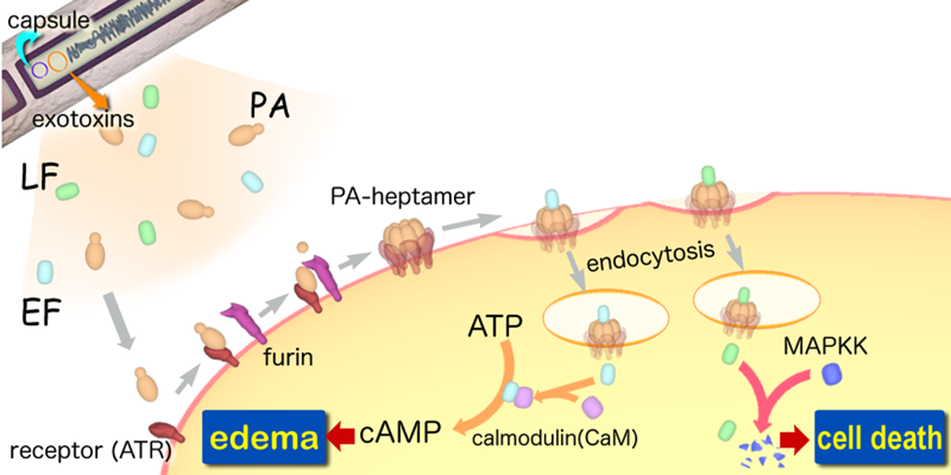
A-B toxin are two-component exotoxin secreted by a number of pathogenic bacteria. The complexes contain two subunits called A subunit and B subunit. A subunit is the active portion that is poisonous to host cells, and is transferred to the host cell through endosomes mediated by the B subunit. The B subunit is a translocation and binding protein. Figure 3 shows how Anthrax toxin enter into human cells (Source: Wikipedia; Author: Y tambe).
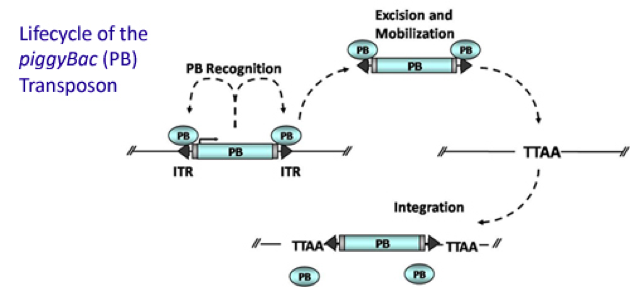
PiggyBac (PB) transposon system
PiggyBac (PB) transposon is a mobile genetic element that efficiently transposes between vectors and chromosomes via a "cut and paste" mechanism but leave no “footprint”. PBase in this system recognizes the transposon-specific inverted terminal repeat sequence (PB5 & PB3 in Figure 6) located on both end of transposon vector and efficiently moves the contents from the original sites and efficiently integrates them into TTAA chromosomal sites. Figure 4 demonstrates the working mechanism of PB transposon system (Source: Wikipedia; Author: Transposagenbio).
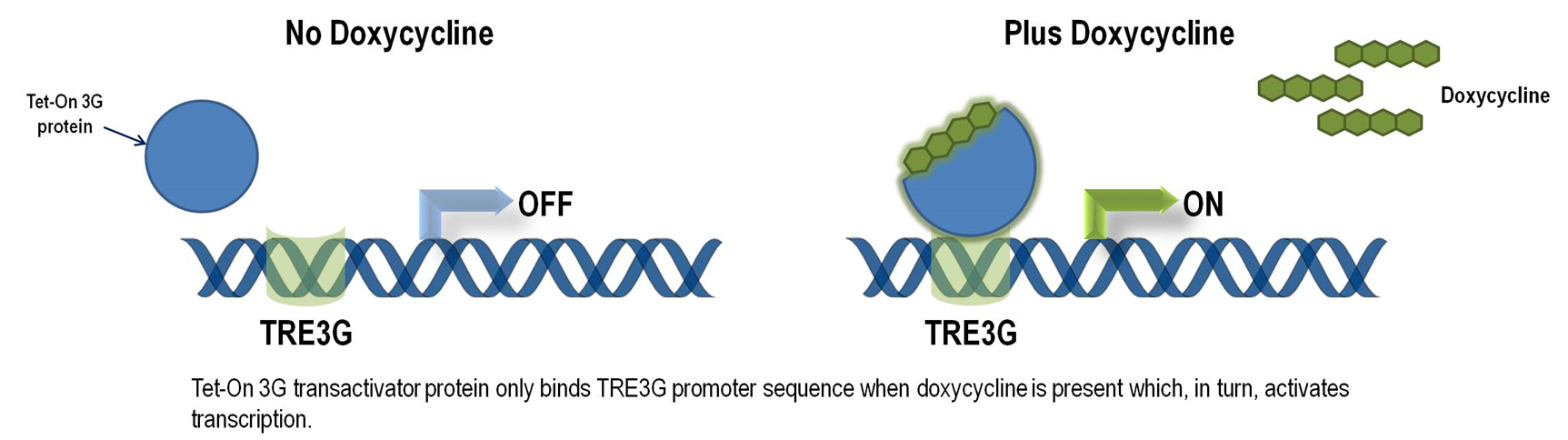
Tet-ON 3G operon system
The Tet-On 3G Systems are inducible gene expression systems for mammalian cells. Target cells that express the Tet-On 3G transactivator protein and contain a gene of interest (GOI) under the control of a TRE3G promoter (PTRE3G) will express high levels of your GOI, but only when cultured in the presence of doxycycline (Dox), which is a synthetic tetracycline derivative. The TRE3G promoter is very sensitive to the concentration of doxycycline and functions like a step function after induction. Also, the Dox concentrations required for induction of Tet-On Systems are far below cytotoxic levels for either cell culture or transgenic studies. So it is ideal to use Tet-ON 3G as a mammalian gene expression controller. Figure 5 demonstrates the mechanism of Tet-ON 3G system (Source: Clontech)
Experiment design
Written by Lin Le, Sijia Liu, Mengqi Xu, Yongkang Long & Yicong Tao
Constructing stable CRISPR/Cas system in human cells
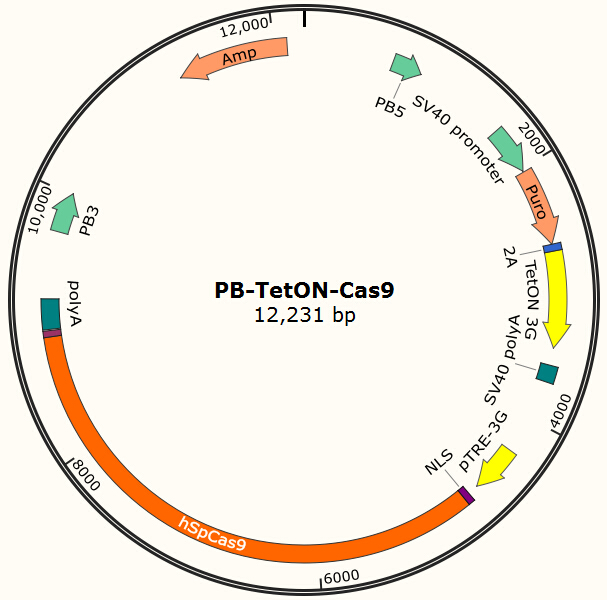
The program aims to endow the human cell the capacity of resisting the infection of HIV by introducing the pruned CRISPR/Cas9 system into the human cell. To wrestle with the high mutation rate of HIV and improve the flexibility of the system, we use PiggyBac transposon to transfect hematopoietic stem cells for permanently and stably expressing Cas9 protein such that all helper T cells differentiate from modified hematopoietic stem cell have ability to defeat HIV by gRNA which is transfected into the cells in need. The DNA segment expressing Cas 9 from pX-330 is cloned into the PiggyBac transposon vector (PB-TetOn-Cas9, Figure 6) which contains a puromycin resistance gene for selection. Cas9 could potentially cause host genome mutagenesis and chromosomal disorders, cytotoxicity, genotoxicity, or oncogenesis. So we use Tet-on 3G operon which is induced by doxycycline to efficiently control the expression of Cas9 in eukaryotic cells and fortify the safety of the system.
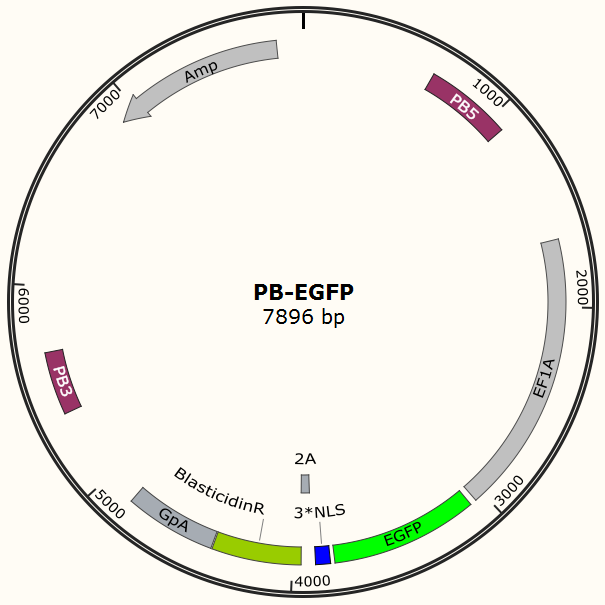
To test the efficiency of the Cas9 and explore the optimal conditions of the system, we need a reporter to indicate the whether the Cas9 functions and the targeted sites are cut off and destructed. So a cell line integrated with EGFP gene is firstly constructed in a similar way. This time the PB-EGFP plasmid (Figure 7), a transposon vector with EGFP gene between the transposon-specific inverted terminal repeat sequences and PiggyBac transposase is co-transfected into cells.
After transfection we treat cell with blasticidin to get a stable cell line expressing EGFP. Then we transiently transfect the cell line with pX330 embedded with gRNA for EGFP and test the efficiency of Cas9-gRNA system.
Deliver gRNA-expressing plasmids by A-B toxin
We choose a non-viral DNA delivery system which is based on modified A-B toxin and yeast GAL4 transcription factor. Yeast transcriptional activator GAL4 protein can specifically recognize plasmids containing UAS sequence. By replacing A subunit of A-B toxin with GAL4, we can get a chimeric fusion protein that can transfer plasmids with UAS into host cells. We construct a plasmid which contains UAS sequence and can transcribe gRNA in human cells, and use it as our gRNA expression vector in human cells.
DNA delivery shuttle design
The chimeric fusion protein mainly comprises 3 parts: target cell-specific binding domain, a translocation domain and a nucleic acid binding domain. The target cell-specific binding domain recognizes the EGF (epidermal growth factor) receptors on the cell surface. The translocation domain enhances nucleic acid escape from the cellular vesicle system and thus to augment nucleic acid transfer. The nucleic acid binding domain, which derives from the yeast GAL4 transcription factor, can carry plasmids with UAS (Upstream Activation Sequence) sequences into cells in vivo. Several research groups from Germany to Taiwan (Gaur, Gupta, Goyal, Wels, & Singh, 2002) (Chen et al., 2000) (Fominaya, Uherek, & Wels, 1998) have achieved this goal. We are requesting the plasmid encoding TEG vehicle from a Germany research group (Wels, Winfried 63110 Rodgau (DE)). Figure 8 shows the schematic representation of the TEG fusion gene in the E.coli expression plasmid pWF47-TEG.
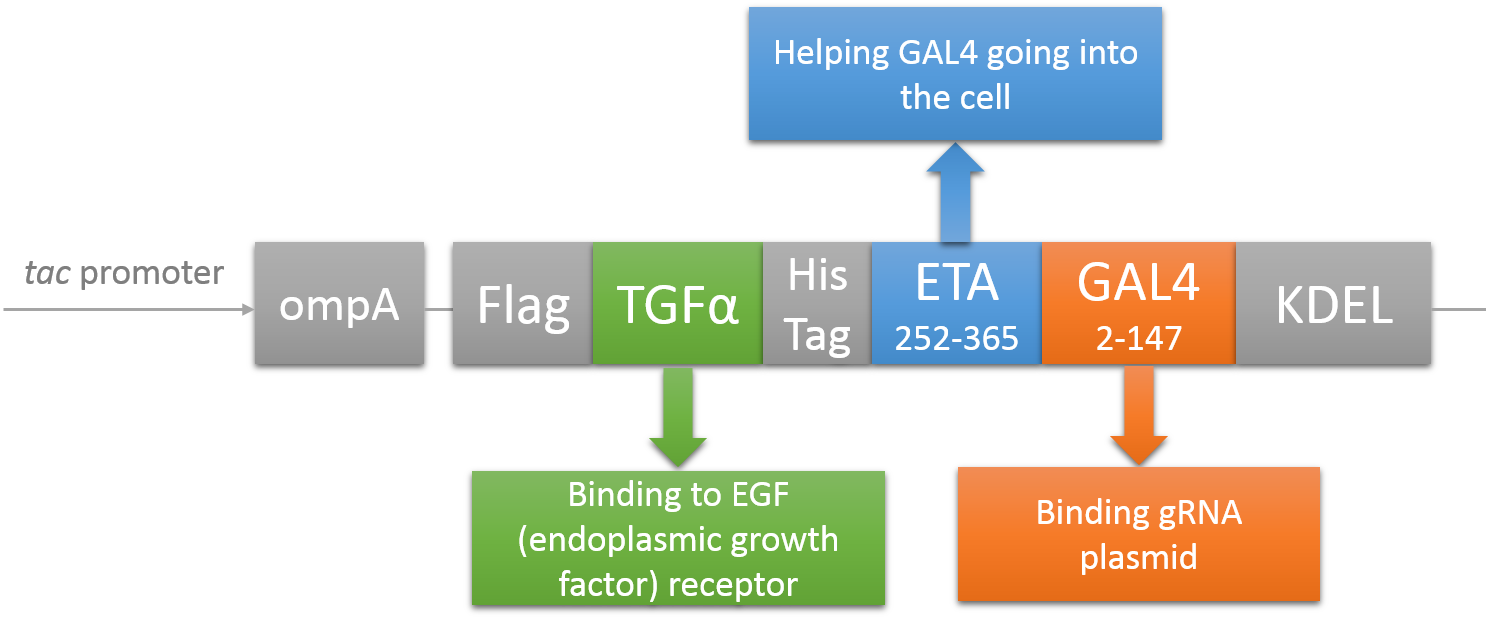
Plasmid & experiment design
Before using this delivery system, we need to check the delivery efficiency of this protein system at first. So, firstly, we construct a plasmid that carries a sgRNA for EGFP, several UAS sequences which are necessary for the vehicle’s recognition and binding and a mCherry gene sequence followed by a NLS (nucleus leading sequence). Figure 9 demonstrates the plasmid we construct.
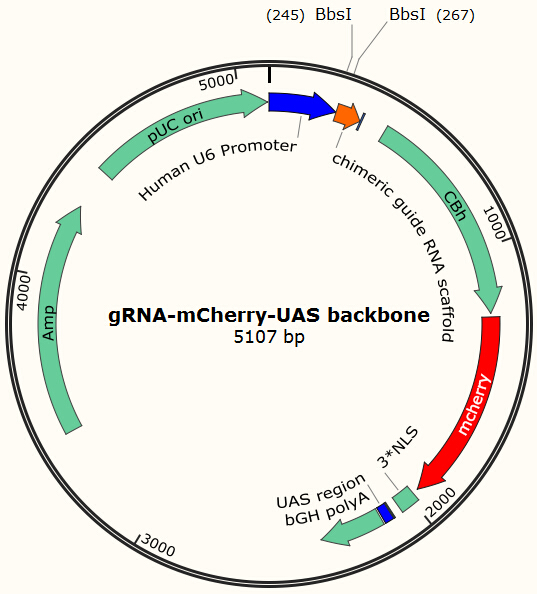
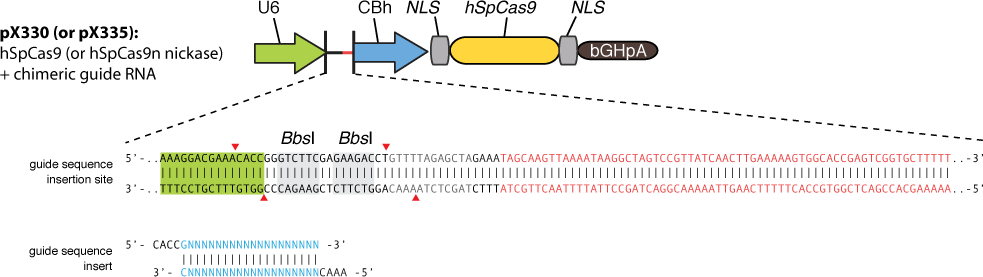
The construction is based on the plasmid backbone of Zhang Lab’s pX330. There are two BbsI restriction enzyme sites between U6 strong promoter and U6 terminator in which we can insert sgRNA. Once sgRNA is inserted, the two BbsI sites disappear and the sgRNA can’t be changed anymore. Figure 10 shows how sgRNA replacement is blocked once BbsI sites are used.
CBh is a eukaryotic promoter, followed by mCherry and 3*NLS. NLS makes red fluorescent protein enter into cell nucleus after expression. By means of PCR and molecular cloning, we can change the number of UAS, a sequence of 17bp which is recognized and bound by GAL4 protein. The sgRNA we used in this plasmid is for EGFP, to collaborate with the Cas9 system and EGFP gene that we have integrated into Hela cell line. In this way, we can visualize how many plasmids are transformed into nucleus by detecting the quantity of red-fluorescence-emitting cells. And with the ratio of reduced green fluorescence and the quantity of red fluorescence we can roughly obtain the efficiency of the CRISPR-Cas system we created. And finally, we reconstruct the plasmid but this time with the sgRNA targeting HIV and test its efficacy.
gRNA design and Modeling
Written by Rongpeng Li and Fan Jiang
gRNA design
gRNA is a 23nt short RNA fragment (not include tracrRNA) which binds to its complementary target and 'guide' Cas9 to cut the target DNA. Since we would like to destroy the HIV virus inside human body, a gRNA sequence that matched a part of HIV conserved region best but have few off-target matches in the human genome is desired. However, designing a gRNA for a human-infective virus is difficult due to the very large difference in the genome size. For virus like HIV, it is even more difficult because HIV can mutate in a very high rate. So finding a perfect gRNA is not realistic. However, finding a group of specific quasi-conservative sequences (gRNA) which are able to target one or more species of HIV is possible.
Our search for the sequence roughly followed the process described in George M. Church's paper (Mali, Yang, et al., 2013). We modified it slightly to fit our own purpose. We first used bioinformatics method to find the quasi-conservative regions in the HIV-1 whole genome reference library by NIH. From the candidates, we selected the region around 720bp from beginning of the genome (aligned), in the less-selected region slightly off the LTR.
We then used the online tools of Feng Zhang's Lab at MIT to find our desired gRNA composition. We also did some calculation based on phenomenological energy calculation to estimate the stability and effectiveness of our gRNA sequences (Hsu et al., 2013). The tool shows that almost no off-target binding will occur. BLAST was used to further confirm the results. We also analyzed the structure of the resulted gRNA, which shows an approximate free energy of -1.4kJ (Zuker, 2003).
For detailed information, see our Modeling page.
Modeling
本段需要重写
The last part is a maverick design which models the inter-cell interaction of T cells to simulate the cell swarm behaviors, our ultimate goal is to determine the desired ratio of cured T cell to recover from a HIV virus carrier.
Our last part focuses on the modeling of the cell interaction. We want to extract a mathematical critical ratio of restored T cells if we want a HIV carrier to be cured. We attempt to apply graph theory and Particle Swarm Optimization to tackle this problem.
Future applications
Written by Yicong Tao
Stably transfect hematopoietic stem cells
August PNAS reveals the possibility of gene therapy in retrovirus diseases
Using CRISPR/Cas to stably transfect itself in a safe locus instead of using Piggybac
In vivo verification
The project is intended to treat retrovirus diseases. Let’s take AIDS as an example to see what’s going to happen. First, stably transfect hematopoietic stem cells with the plasmid which encodes Cas9 protein. Second, we use bioinformatics to screen out a 20bp conserved regions in HIV DNA which has no conservation in human genome. Then, constructing a plasmid which encodes the gRNA sequence. When the human body is infected by AIDS, using non-viral DNA transfecting methods (e.g. A-B toxin based shuttle) to deliver the plasmid into human somatic cells (especially CD4+ cells) and the CRISPR/Cas system is activated. The Cas9 protein recognizes viral DNA with the help of gRNA and cut the target sequence, causing double strand break (DSB). The DSB is then repaired by the non-homologous end joining mechanism in the cell but with high error rate, causing frame-shift mutation and gene knockout. The viral protein synthesis is stopped and further infection is impossible, providing potentiation for eradicating AIDS.
References
Chen, T. Y., Hsu, C. T., Chang, K. H., Ting, C. Y., Whang-Peng, J., Hui, C. F., & Hwang, J. (2000). Development of DNA delivery system using Pseudomonas exotoxin A and a DNA binding region of human DNA topoisomerase I. Applied Microbiology and Biotechnology, 53(5), 558-567.
Cong, L., Ran, F. A., Cox, D., Lin, S. L., Barretto, R., Habib, N., . . . Zhang, F. (2013). Multiplex Genome Engineering Using CRISPR/Cas Systems. Science, 339(6121), 819-823. doi: DOI 10.1126/science.1231143
Fominaya, J., Uherek, C., & Wels, W. (1998). A chimeric fusion protein containing transforming growth factor-alpha mediates gene transfer via binding to the EGF receptor. Gene Ther, 5(4), 521-530. doi: 10.1038/sj.gt.3300614
Gaur, R., Gupta, P. K., Goyal, A., Wels, W., & Singh, Y. (2002). Delivery of nucleic acid into mammalian cells by anthrax toxin. Biochemical and Biophysical Research Communications, 297(5), 1121-1127. doi: Pii S0006-291x(02)02299-4 Doi 10.1016/S0006-291x(02)02299-4
Hsu, P. D., Scott, D. A., Weinstein, J. A., Ran, F. A., Konermann, S., Agarwala, V., . . . Zhang, F. (2013). DNA targeting specificity of RNA-guided Cas9 nucleases. Nature Biotechnology, 31(9), 827-+. doi: Doi 10.1038/Nbt.2647
Mali, P., Esvelt, K. M., & Church, G. M. (2013). Cas9 as a versatile tool for engineering biology. Nat Methods, 10(10), 957-963. doi: 10.1038/nmeth.2649
Mali, P., Yang, L., Esvelt, K. M., Aach, J., Guell, M., DiCarlo, J. E., . . . Church, G. M. (2013). RNA-guided human genome engineering via Cas9. Science, 339(6121), 823-826. doi: 10.1126/science.1232033
Zuker, M. (2003). Mfold web server for nucleic acid folding and hybridization prediction. Nucleic Acids Res, 31(13), 3406-3415. doi: Doi 10.1093/Nar/Gkg595
 "
"