Team:HNU China/Project
From 2014.igem.org
(Difference between revisions)
Yannickchou (Talk | contribs) |
Yannickchou (Talk | contribs) |
||
| Line 268: | Line 268: | ||
</div> | </div> | ||
<div class="wrapper"> | <div class="wrapper"> | ||
| - | <div class="tabset" id="tabset1"><a id="a" class="tab sel {content:'cont_1'}"><b>Background</b></a><a id="b" class="tab {content:'cont_2'}"><b>Description</b></a><a id="c" class="tab {content:'cont_3'}"><b> | + | <div class="tabset" id="tabset1"><a id="a" class="tab sel {content:'cont_1'}"><b>Background</b></a><a id="b" class="tab {content:'cont_2'}"><b>Description</b></a><a id="c" class="tab {content:'cont_3'}"><b>Experiment</b></a><a id="d" class="tab {content:'cont_4'}" ><b>Result</b></a><a id="a" class="tab {content:'cont_5'}" ><b>Future work</b></a></div> |
<div id="cont_1"> | <div id="cont_1"> | ||
<div id="chead"> | <div id="chead"> | ||
| Line 436: | Line 436: | ||
<div id="cont_3"> | <div id="cont_3"> | ||
<div id="chead"> | <div id="chead"> | ||
| - | <div align="left"class="STYLE3" style="margin-left:50px;"><br/><br/><br/> | + | <div align="left"class="STYLE3" style="margin-left:50px;"><br/><br/><br/>Experiment</div> |
</div> | </div> | ||
<div id="cmiddle"> | <div id="cmiddle"> | ||
| Line 443: | Line 443: | ||
Preliminary construction of the Cry2-CIB1 system <br/><br/> | Preliminary construction of the Cry2-CIB1 system <br/><br/> | ||
| - | <b>I. | + | <b>I.Cloning of the Cry2 & CIB1 gene</b> <br/><br/> |
| - | + | According to the sequences from NCBI, we designed two pairs of primers respectively, and cloned them based on the cDNA library of <i>Arabidopsis</i>. To fulfill the requirement of following gateway operation, the attB1/2 are added to the primers.<br/><br/> | |
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | According to the sequences from NCBI, we designed two pairs of primers respectively, and cloned them based on the cDNA library of <i>Arabidopsis</i>. | + | |
Primer for Cry2: <br/> | Primer for Cry2: <br/> | ||
| - | Fwd 5’→3’ ATGAATGGAGCTATAGGA <br/> | + | Fwd 5’→3’ <span class="STYLE4">GTACAAAAAAGCAG</span>ATGAATGGAGCTATAGGA <br/> |
| - | Rev 5’→3’ TCAAACTCCTAAATTGCC <br/><br/> | + | attB1 <br/> |
| + | Rev 5’→3’ <span class="STYLE4">GTACAAGAAAGCTGGGT</span>TCAAACTCCTAAATTGCC <br/><br/> | ||
| + | attB2 <br/><br/> | ||
Primer for CIB1: <br/> | Primer for CIB1: <br/> | ||
| - | Fwd 5’→3’ ATGAAGATGGACAAAAAGA <br/> | + | Fwd 5’→3’ <span class="STYLE4">GTACAAAAAAGCAG</span>ATGAAGATGGACAAAAAGA <br/> |
| - | Rev 5’→3’ TCATTTGCAACCATTTTT <br/><br/> | + | attB1 <br/> |
| + | Rev 5’→3’ <span class="STYLE4">GTACAAGAAAGCTGGGT</span>TCATTTGCAACCATTTTT <br/><br/> | ||
| + | attB1 <br/> | ||
| - | <b> | + | <b>II.Yeast-two-hybrid </b> <br/><br/> |
| - | + | What we chose is the commercial shuttle vectors, pDEST32 and pDEST22[fig.1], where the DBD and AD is actually Gal4 BD and Gal4 AD respectively, and the Gal4 AD shall specifically bind to the upstream activating sequence(UAS), locating in the promoters of several reporter genes in the yeast strain genome like lacZ, HIS3 as well URA3.<br/><br/> | |
| - | What we chose is the pDEST32 and pDEST22[fig. | + | |
| - | Fig.2 <br/> | + | Fig.2 The map of our two plasmids: pDEST22 and pDEST32<br/> |
<img src="https://static.igem.org/mediawiki/2014/0/09/Experimentation_fig2.jpg" /><br/><br/> | <img src="https://static.igem.org/mediawiki/2014/0/09/Experimentation_fig2.jpg" /><br/><br/> | ||
| - | Since we have obtained CRY2 and CIB1, what we do next is to induce both of them into their vectors with the technology named Gateway, constructing two new plasmids, | + | Since we have obtained CRY2 and CIB1, what we do next is to induce both of them into their vectors with the technology named Gateway, constructing two new plasmids, pDEST22-CIB1 and pDEST32-32 as below[fig.2]. Then we transform the plasmids above into the Saccharomyces cerevisiae cell, AH109, screening by Leu and Trp auxotroph. <br/><br/> |
| - | pDEST22-CIB1 and pDEST32-32 as below[fig. | + | |
| - | Fig.3 <br/> | + | Fig.3 The map of our two plasmids: pDEST22-CIB1 and pDEST32-CRY2<br/> |
<img src="https://static.igem.org/mediawiki/2014/d/d9/Experimentation_fig3.jpg" /><br/><br/> | <img src="https://static.igem.org/mediawiki/2014/d/d9/Experimentation_fig3.jpg" /><br/><br/> | ||
| - | + | <b>III. Optogenetic test</b> <br/><br/> | |
| - | + | To test the effect of light switchable gene expression system, blue-white selection is induced when the blue light in special incubator[fig.3] shall initiate the expression of LacZ, which combined with the IPTG and X-gal in solid medium, makes the colony into blue, contrarily, colony in dark remains white.[fig.4]<br/><br/> | |
| + | |||
| + | Fig.3 Blue light incubator<br/> | ||
<img src="https://static.igem.org/mediawiki/2014/1/19/Experimentation_fig4.jpg" /><br/><br/> | <img src="https://static.igem.org/mediawiki/2014/1/19/Experimentation_fig4.jpg" /><br/><br/> | ||
| - | + | Fig.4 The positive result of yeast X-gal coloration | |
| - | < | + | <img src="https://static.igem.org/mediawiki/2014/b/b6/Exfig4.jpg" /><br/><br/> |
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | <br/> | + | |
<img src="https://static.igem.org/mediawiki/2014/7/73/July.png" width="98" height="45" /> <br/> | <img src="https://static.igem.org/mediawiki/2014/7/73/July.png" width="98" height="45" /> <br/> | ||
| - | Cloning of the genes we need and | + | Cloning of the genes we need and ligated them into of T-vector <br/><br/> |
<b>I.IRE-IRP</b> <br/><br/> | <b>I.IRE-IRP</b> <br/><br/> | ||
| - | |||
| - | |||
1.IRP <br/><br/> | 1.IRP <br/><br/> | ||
| Line 501: | Line 492: | ||
We decide to clone the IRP from human cDNA library. <br/><br/> | We decide to clone the IRP from human cDNA library. <br/><br/> | ||
| - | Primers for IRP[fig. | + | Primers for IRP[fig.5]: <br/> |
Fwd 5’→3’ GCG<span class="STYLE4">AAGCTT</span>TCAGTAATCATGAGCAAC <br/> | Fwd 5’→3’ GCG<span class="STYLE4">AAGCTT</span>TCAGTAATCATGAGCAAC <br/> | ||
HindIII <br/> | HindIII <br/> | ||
| Line 507: | Line 498: | ||
SacI <br/><br/> | SacI <br/><br/> | ||
| - | Fig. | + | Fig.5<br/> |
<img src="https://static.igem.org/mediawiki/2014/4/4b/Jiao1.jpg" /><br/><br/> | <img src="https://static.igem.org/mediawiki/2014/4/4b/Jiao1.jpg" /><br/><br/> | ||
| Line 522: | Line 513: | ||
<b>II.FET3</b> <br/><br/> | <b>II.FET3</b> <br/><br/> | ||
| - | + | 1.Prepare for the genome of <i>Saccharomyces cerevisiae</i> <br/><br/> | |
| - | + | ||
| - | + | ||
Due to the lack of existing yeast genome as the PCR template, we order a yeast genome extraction kit to prepare the source.[fig.2] <br/><br/> | Due to the lack of existing yeast genome as the PCR template, we order a yeast genome extraction kit to prepare the source.[fig.2] <br/><br/> | ||
| Line 557: | Line 546: | ||
<img src="https://static.igem.org/mediawiki/2014/1/18/Jiao4.jpg" /><br/><br/> | <img src="https://static.igem.org/mediawiki/2014/1/18/Jiao4.jpg" /><br/><br/> | ||
| - | <b>IV. | + | <b>IV.TA cloning</b> <br/><br/> |
To make sure the PCR-cloned sequences are expected, we ligate them into the pMD18-T vector, then sequence all together. On the other hand, objective sequence in circular plasmids makes it convenient and stable for storage for quite long time. <br/><br/> | To make sure the PCR-cloned sequences are expected, we ligate them into the pMD18-T vector, then sequence all together. On the other hand, objective sequence in circular plasmids makes it convenient and stable for storage for quite long time. <br/><br/> | ||
| - | < | + | <img src="https://static.igem.org/mediawiki/2014/a/a0/August.png" width="98" height="45" /> <br/> |
| - | + | ||
| - | + | Construction of the two plasmids in iron sensitive absorbing system<br/><br/> | |
| - | + | ||
| - | + | Since we comfronted trouble in the ligation of gene with shuttle vector by T4 ligase after restriction enzyme treatment, the schematics below is just our original plan.<br/><br/> | |
| - | + | ||
| - | + | <b>I.pDEST22-IRP1</b> <br/><br/> | |
| - | <br/> | + | |
| + | Concerning the limitation of restriction cloning site, We have to ligated IRP1 firstly with pDEST32, and finally tranfer it into the pDEST22 vector.<br/><br/> | ||
| + | |||
| + | 1.When pDEST32 are treated with HindIII SacI, it yields segment of 2.3K & 10K.<br/> | ||
| + | 2.When T-IRP1 are treated with HindIII SacI, it yields segment of 2.6K.<br/> | ||
| + | 3.We can ligate the 10K and 2.6K segments together, constructing pDEST32-IREBP1.<br/> | ||
| + | 4.Restriction enzymens XhoI and SacI digest pDEST22 and pDEST32-IREBP1 respectively. <br/> | ||
| + | 5.The pDEST22-IREBP1 comes from the ligation of 5.1K segment from pDEST22 yields and the 4.1K segment from pDEST32-IREBP1 yields.<br/><br/> | ||
| + | |||
| + | <b>II.pDEST32-IREBP1</b> <br/><br/> | ||
| + | |||
| + | 1.Restriction enzymens HindIII and SacI digest pDEST32 and T-IRE respectively. <br/> | ||
| + | 2.pDEST32-IRE is the ligation product of 10K segment from pDEST32 and IRE<br/> | ||
| + | 3.Restriction enzymens SpeI and SacI digest pDEST32-IRE and T-FET3 respectively. <br/> | ||
| + | 4.pDEST32-IRE-FET3 is the ligation product of 10K segment from pDEST32-IRE and 2K segment from T-FET3<br/> | ||
</div> | </div> | ||
Revision as of 01:21, 18 October 2014
Background
Introduction
Biomining, is used to describe the novel approach in mining industry when microorganisms are applied to the extraction and recovery of precious and base metals from ores and concentrates[fig.1]. Technicially, it consists two branch, the bioleaching and biooxidation. The bioleaching strictly refers to the case when microorganisms are used to solubilize the metal. While biooxidation maily focus on the pretreatment of target metals by bio-processsing, mening minerals that occlude target metals, such as thiosulfate encompasses the gold.

Fig.1 Biomining is used to extract copper from copper ore.
In reality, the same biological process had been unknowingly used to extract metals at mine sites in, for example Spain, the UK and China, for several hundred years. The modern era of bioming began with the discovery of the bactreium, Thiobacillus ferrooxidans(now Acidithiobacillus ferrooxidans) in the mid-1940s and the initial understanding of this microbe’s involvement in copper extraction. In 1958 Kennecott Mining Company patented the use of Thiobacillus ferrooxidans for copper extraction from waste rock dumps at the Bingham Canyon mine in Utah, USA.As a survey illustrates that currently bioming is commercially praticed in the production of 15% of copper, 5% of gold.
Why biomine?
Bio-extractive techniques have to compete with alternative approaches for extracting metals from ores and concentrates. Some, such as pyrometallurgical technologies[fig.2] (ore roasting/smelting) have been refined over millennia and often represent major investments by mining companies, while others, such as pressure leaching, are more recent non-biological innovations. Main microorganisms involved in mineral oxidation processes are autotrophs, and the processes operate usually at atmospheric pressure and at relatively low temperatures (20–80 8C). Biomining is generally perceived as a much more environmentally benign (‘green’) approach, involving much lower temperatures (and hence energy costs) and smaller carbon footprints, which contrasts with current biomining operations, relied on the blasting and grinding of ore bodies, emitting large amounts of CO2, consuming 5% of total global energy production. Bio- processing also has niche advantages where firstly, the ore or concentrate contains significant quantities of arsenic, and secondly, for processing lowgrade and complex (polymetallic) ores.

Fig2. Pyrometallurgy
Configurations and microbiology for biomining
Engineering options for biomining have evolved from relatively inexpensive, partly controlled, irrigated dump or heap reactors to sophisticated, highly controlled and expensive stirred-tank reactors. Another distinguished way of bioming is the in situ mining, used extensively in Canada in the 1970s to recover uranium from worked out deep mines, which may well be the next major development in the mining sector. Mineral heaps and stirred tanks provide very different environments and challenges for mineral-leaching microorganisms, and different ‘optimal’ populations might be expected to emerge with similar target minerals depending on the reactor used.
The extreme physico-chemical nature of bioleach liquors — low pH, elevated concentrations of (toxic) metals, metalloids and other solutes, and highly positive redox potential (EH values may exceed +900 mV) — means that they are highly toxic to the vast majority of life forms, including microorganisms. Microorganisms oxidize both sulfur and iron of sulfide minerals, such as pyrite. It is now well established that bioleaching and biooxidation in all biomining operations is mediated by consortia of acidophilic prokaryotes. These have been categorized as: firstly, ferric iron-generating autotrophs which produce the mineral oxidant; secondly, sulfuric acid-generating autotrophs, which maintain the low pH environment required; and finally, heterotrophic and mixotropic prokaryotes, which degrade organic compounds leaked from autotrophic iron-oxidizers and sulfur-oxidizers, there avoiding potential toxicity issues.
Engineered microbial consortia
The field of synthetic biology has developed a wide range of highly engineered clonal populations of bacteria to perform complex tasks.The construction and analysis of synthetic gene circuits has not only provided us with new tools for genetic engineering but has given deeper insight into naturally occurring gene circuits, their evolution, architec.tures, and properties as well. The industrial practice of biomining and bioremediation of heavy metal contaminations could potentially benefit from synthetic consortia as natural consortia have been shown to play crucial roles in these processes.
To our best knowledge, hybrid consortia consisting of genetically engineered and naturally occurring bioleaching bacteria have not been reported so far. In fact only two knockouts and two expression mutants have been reported in the scientific literature. One rus overexpressing A. ferrooxidans strain and another expressing the mer determinant for a mercury resistant A. caldus strain. Once more suitable transformation protocols have been developed, it maybe feasible to modulate quorum-sensing signals with engineered microbes by either attenuating or amplifying natural signals or sending artificial signals to promote biofilm formation or mobilization respectively as recently demonstrated with engineered E. coli cells.
Fungi in acid mine drainage
Roles of fungi, the natural residents of acid mine drainage(AMD) and its attenuator are not emphasized adequately in the mine water research. Though AMD appears to be a certain special enviroment with relatively high metal concentration as well low pH, several species of fungi are also isolated from the AMD carrying streams, even AMD. Generally, fungi occur over a wide pH range (pH 1.0–11.0) and have been detected in acid habitats like volcanic springs, acid mine drainage or acid industrial wastewaters. Many of them are primarily acid-tolerant, but truly acidophilic species have also been detected[talbe.1]. While extracellular precipitation, complexation and crystallization, metal transformation, biosorption and sequestration are seen for fungi, endow fungi the ability to survive.

Under this condition, fungi may play the important role as primary degraders of complex organic matter, due to the absence of invertebrates that actively shred the leaves (shredders) at pH values below 3.5. At the same time, the fungi will contribute to oxygen consumption, thereby limiting oxidative stress for the anaerobic bactiria. Moreover, fungi can be directly involved in the reduction of ferric iron or sulphur. Comparatively, more reports are available concerning the absorption of heavy metals by fungi, in comparison to bacteria or algae in freshwater ecosystem. Fungi can absorb metals in their cell wall or adsorb in extracellular polysaccharide slime. This capacity enables them to grow in the presence of high amounts of heavy metals. Fungal activity in acid mine drainage is represented in Fig.3

Fig.3 Schematic diagram of fungal influence in acid mine drainage remediation
Conclusion
Biomining, is used to describe the novel approach in mining industry when microorganisms are applied to the extraction and recovery of precious and base metals from ores and concentrates. Compared with the conventional approachs, such as pyrometallurgical technologies and pressure leaching in mining industry, bioming has its unique advantages in energy conservation and emission reduction, both of which are definitely with rising importance in the modern time. Microorganisms are usually settled in the non-sterile condition, and participate the bio-processing in consortia. In most of the time, they play the role of iron-oxidizer, sulfur-oxdizer and mixotrophic or heterotrophic acidophiles repectively or together. In the “bottom up” approach to optimize the consortia, the logic of synthetic biology is a worthy try to get the optimum colonies, though short of precedent. Fungi is a natural resident in the acid mine drainage and involved in the reduction of ferric iron and sulphur, matching the mainly role in bioming consortia. Since Saccharomyces cerevisiae is one of the model organisms people study in, scientists has developed a relatively mature genetically engineering operation. According to above reasons, we chose Saccharomyces cerevisiae as a model, aiming to optimize the ability as a biomining microorganism with our biobricks.
Reference
Biomining, is used to describe the novel approach in mining industry when microorganisms are applied to the extraction and recovery of precious and base metals from ores and concentrates[fig.1]. Technicially, it consists two branch, the bioleaching and biooxidation. The bioleaching strictly refers to the case when microorganisms are used to solubilize the metal. While biooxidation maily focus on the pretreatment of target metals by bio-processsing, mening minerals that occlude target metals, such as thiosulfate encompasses the gold.

Fig.1 Biomining is used to extract copper from copper ore.
In reality, the same biological process had been unknowingly used to extract metals at mine sites in, for example Spain, the UK and China, for several hundred years. The modern era of bioming began with the discovery of the bactreium, Thiobacillus ferrooxidans(now Acidithiobacillus ferrooxidans) in the mid-1940s and the initial understanding of this microbe’s involvement in copper extraction. In 1958 Kennecott Mining Company patented the use of Thiobacillus ferrooxidans for copper extraction from waste rock dumps at the Bingham Canyon mine in Utah, USA.As a survey illustrates that currently bioming is commercially praticed in the production of 15% of copper, 5% of gold.
Why biomine?
Bio-extractive techniques have to compete with alternative approaches for extracting metals from ores and concentrates. Some, such as pyrometallurgical technologies[fig.2] (ore roasting/smelting) have been refined over millennia and often represent major investments by mining companies, while others, such as pressure leaching, are more recent non-biological innovations. Main microorganisms involved in mineral oxidation processes are autotrophs, and the processes operate usually at atmospheric pressure and at relatively low temperatures (20–80 8C). Biomining is generally perceived as a much more environmentally benign (‘green’) approach, involving much lower temperatures (and hence energy costs) and smaller carbon footprints, which contrasts with current biomining operations, relied on the blasting and grinding of ore bodies, emitting large amounts of CO2, consuming 5% of total global energy production. Bio- processing also has niche advantages where firstly, the ore or concentrate contains significant quantities of arsenic, and secondly, for processing lowgrade and complex (polymetallic) ores.

Fig2. Pyrometallurgy
Configurations and microbiology for biomining
Engineering options for biomining have evolved from relatively inexpensive, partly controlled, irrigated dump or heap reactors to sophisticated, highly controlled and expensive stirred-tank reactors. Another distinguished way of bioming is the in situ mining, used extensively in Canada in the 1970s to recover uranium from worked out deep mines, which may well be the next major development in the mining sector. Mineral heaps and stirred tanks provide very different environments and challenges for mineral-leaching microorganisms, and different ‘optimal’ populations might be expected to emerge with similar target minerals depending on the reactor used.
The extreme physico-chemical nature of bioleach liquors — low pH, elevated concentrations of (toxic) metals, metalloids and other solutes, and highly positive redox potential (EH values may exceed +900 mV) — means that they are highly toxic to the vast majority of life forms, including microorganisms. Microorganisms oxidize both sulfur and iron of sulfide minerals, such as pyrite. It is now well established that bioleaching and biooxidation in all biomining operations is mediated by consortia of acidophilic prokaryotes. These have been categorized as: firstly, ferric iron-generating autotrophs which produce the mineral oxidant; secondly, sulfuric acid-generating autotrophs, which maintain the low pH environment required; and finally, heterotrophic and mixotropic prokaryotes, which degrade organic compounds leaked from autotrophic iron-oxidizers and sulfur-oxidizers, there avoiding potential toxicity issues.
Engineered microbial consortia
The field of synthetic biology has developed a wide range of highly engineered clonal populations of bacteria to perform complex tasks.The construction and analysis of synthetic gene circuits has not only provided us with new tools for genetic engineering but has given deeper insight into naturally occurring gene circuits, their evolution, architec.tures, and properties as well. The industrial practice of biomining and bioremediation of heavy metal contaminations could potentially benefit from synthetic consortia as natural consortia have been shown to play crucial roles in these processes.
To our best knowledge, hybrid consortia consisting of genetically engineered and naturally occurring bioleaching bacteria have not been reported so far. In fact only two knockouts and two expression mutants have been reported in the scientific literature. One rus overexpressing A. ferrooxidans strain and another expressing the mer determinant for a mercury resistant A. caldus strain. Once more suitable transformation protocols have been developed, it maybe feasible to modulate quorum-sensing signals with engineered microbes by either attenuating or amplifying natural signals or sending artificial signals to promote biofilm formation or mobilization respectively as recently demonstrated with engineered E. coli cells.
Fungi in acid mine drainage
Roles of fungi, the natural residents of acid mine drainage(AMD) and its attenuator are not emphasized adequately in the mine water research. Though AMD appears to be a certain special enviroment with relatively high metal concentration as well low pH, several species of fungi are also isolated from the AMD carrying streams, even AMD. Generally, fungi occur over a wide pH range (pH 1.0–11.0) and have been detected in acid habitats like volcanic springs, acid mine drainage or acid industrial wastewaters. Many of them are primarily acid-tolerant, but truly acidophilic species have also been detected[talbe.1]. While extracellular precipitation, complexation and crystallization, metal transformation, biosorption and sequestration are seen for fungi, endow fungi the ability to survive.

Under this condition, fungi may play the important role as primary degraders of complex organic matter, due to the absence of invertebrates that actively shred the leaves (shredders) at pH values below 3.5. At the same time, the fungi will contribute to oxygen consumption, thereby limiting oxidative stress for the anaerobic bactiria. Moreover, fungi can be directly involved in the reduction of ferric iron or sulphur. Comparatively, more reports are available concerning the absorption of heavy metals by fungi, in comparison to bacteria or algae in freshwater ecosystem. Fungi can absorb metals in their cell wall or adsorb in extracellular polysaccharide slime. This capacity enables them to grow in the presence of high amounts of heavy metals. Fungal activity in acid mine drainage is represented in Fig.3

Fig.3 Schematic diagram of fungal influence in acid mine drainage remediation
Conclusion
Biomining, is used to describe the novel approach in mining industry when microorganisms are applied to the extraction and recovery of precious and base metals from ores and concentrates. Compared with the conventional approachs, such as pyrometallurgical technologies and pressure leaching in mining industry, bioming has its unique advantages in energy conservation and emission reduction, both of which are definitely with rising importance in the modern time. Microorganisms are usually settled in the non-sterile condition, and participate the bio-processing in consortia. In most of the time, they play the role of iron-oxidizer, sulfur-oxdizer and mixotrophic or heterotrophic acidophiles repectively or together. In the “bottom up” approach to optimize the consortia, the logic of synthetic biology is a worthy try to get the optimum colonies, though short of precedent. Fungi is a natural resident in the acid mine drainage and involved in the reduction of ferric iron and sulphur, matching the mainly role in bioming consortia. Since Saccharomyces cerevisiae is one of the model organisms people study in, scientists has developed a relatively mature genetically engineering operation. According to above reasons, we chose Saccharomyces cerevisiae as a model, aiming to optimize the ability as a biomining microorganism with our biobricks.
Reference
- Biomining — biotechnologies for extracting and recovering metals from ores and waste materials. D Barrie Johnson. Current Opinion in Biotechnology 2014, 30:24–31
- The microbiology of biomining: development and optimization of mineral-oxidizing microbial consortia. Douglas E. Rawlings1 and D. Barrie Johnson2.Microbiology (2007), 153, 315–324
- How will biomining be applied in future? C. L. BRIERLEY. Trans. Nonferrous Met. Soc. China 18(2008) 1302-1310
- Engineering microbial consortia to enhance biomining and bioremediation. Karl D. Brune andTravis S. Bayer* . Frontiers in Microbiology. 05 June 2012
- Occurrence and role of algae and fungi in acid mine drainage environment with special reference to metals and sulfate immobilization. Bidus Kanti Dasa, Arup Roya, Matthias Koschorreckb, Santi M. Mandalc, Katrin Wendt-Potthoffb, Jayanta Bhattacharyaa,*. water research 43 (2009)883–894
Description
I.Overview
In the mining industry, biomining, with its specific characteristics in energy conservation and emission reduction, is under accelerating development. Microbial consortium used in biomining mainly play the role as iron-oxidizer, sulfur-oxidizer or heterotrophic one. Microorganisms are also reported to be applied in the biomining’s following steps, like the metal recovery from the abandoned ore or slags, and Acid Mine Drainage(AMD) remediation. Fungi are the natural residents of AMD, with the ability of ferric iron oxidation and absorption, can take part into the biomining process and the following metal reduction operation. With the genetically engineering mediation, we may create a new colony of Saccharomyces cerevisiae with intensified potency in industrial practicing. For the biosafety reason, we would attach apoptosis system to ensure the engineering fungus shall not raise biological contamination. Due to the inefficiency in transformaiton and genetic manipulation, the “top down” method are widely adopted in the existing practice, which mesns indigenous microflora are inculated into the reactor and compete with each other. As far we know, It is the fisrt time introduing synthetic biology in biomining optimation in the logic of “bottom up”. The mature genetic manipulation in Saccharomyces cerevisiae ensures the feasibility, and perhaps our biobrick can be used in other organisms in the future, once engineering become facile for the other organisms.
Our microbial miner briefly consists of two systems, iron sensitive absorbing system and optogenetic apoptosis system.
II.Iron sensitive aborbing system
1.FET3 protein
High affinity iron uptake in Saccharomyces cerevisiae requires FET3 protein. Localized on the external cell surface, FET3 protein is proposed to facilitate iron uptake by catalyzing the oxidation of Fe(II) to Fe(III) by O2, shown as below:
4Fe(II) + O2 + 4H+ → 4Fe(III) + 2H2O
The posttranslational insertion of four copper ions into FET3 protein is essential for its activity, thus linking copper and iron homeostasis[fig.1]. In this model, Fe(III) is the substrate for the iron permease, encoded by FTR1 protein[fig2].

Fig.1 Ribbon diagram of FET3 protein. The T1, T2, and T3 copper atoms are shown in blue, green, and yellow, respectively. The three plastocyanin-like domains are shown as blue, green, and gold. Included in red are the carbohydrates resolvable in the electron density map.

Fig.2 FET3 and FTR1 proteins assemble into a complex in a cellular compartment early in the secretory pathway, most likely in the ER. The complex progresses to a post-Golgi compartment, where the CCC2 protein mediates copper delivery to FET3 protein. Finally, the copper-loaded F ET3 protein, presumably still complexed to FTR1 protein, is delivered to the plasma membrane and becomes competent for iron transport.
2.IRE-IRP1
Saccharomyces cerevisiae post-translationally regulates the expression of the plasma membrane high affinity iron transport system, consisting FET3 protein and FTR1 protein, in response to iron need. Incubation of cells in high iron leads to the internalization and degradation of both FET3 protein and FTR1 protein. Since what we expect is a way to accumulate iron that shall promote the absorption in presence of it, we cast our eyes to other mammalian regulation system involving iron.
In human cell, the cellular levels of ferritin and transferrin receptor (TfR) are also primarily regulated at translational level by changes in iron availability, through interactions between iron regulatory proteins (IRP1 and IRP2) and iron responsive elements (IRE) contained within the 5’ UTR of H- and L-ferritin mRNA and the 3’ UTR of TfR mRNA. When intracellular iron levels rise, IRP1 is no longer able to bind IRE, IRP2 is degraded and ferritin mRNA is efficiently translated. [fig.3] Apparently, the FET-IRP1 connetion from ferritin suits our anticipation to open the expression of objective gene. When iron level is high, IRP1 exist as a cytosolic aconitase. When iron islow, it assumes an open configuration associated with the loss of iron atoms in the iron-sulfur cluster, and can bind the IRE stem loop.[fig.4]
Fig.3 Decreased iron supply activates binding of IRPs to IRE resulting in translational inhibition of the mRNAs encoding ferritin and stabilization of the Tf R mRNA. During high iron conditions, IRPs lose their affinity for IREs, increasing translation of ferritin mRNAs and degradation of the Tf R mRNA.

Fig.4 The Fe-S cluster in aconitase.
3.Our project
What we plan is to integrate the FET3 from Saccharomyces cerevisiae with IRE-IRP1 system from human into the fungus, endowing it the ability of iron-sensitively enhanced aborption system. In the circumstance with low iron concentration, the constitutively expressing IRP1 bonds to the stem loop of IRE preceding the FET3 gene, impeding the expression of FET3. Once iron reaches to a high level, the breakdown of IRE-IRP1 connection shall activate the FET3 gene, which lead to more iron uptake, at last achieve the goal for iron accumulation.[fig.5]

Fig.5 Iron sensitive aborbing system
III.Optogenetic apoptosis system
1.Caspase-3
Caspase-3, a member of the caspase family of cell death proteases, cleaves cytoplasmic and nuclear substrates and promotes apoptotic cell death in mammalian cells[fig.6].

Fig.6 Extrinsic and intrinsic pathways to caspase-3 activation.
Although yeasts lack some elements of the complex apoptotic machinery of metazoan cells, recent studies show that many features of apoptosis, including a caspase-like activity, can be induced in these organisms by DNA damage and other apoptotic triggers. Here we plan to introduce CASP3 gene from human, and attempt to verify our hypothesis of the apoptosis effect in yeast by Caspase-3.
2.CRY2-CIB1
Cryptochromes (CRY) are photolyase-like blue-light receptors that mediate light responses in plants and animals. Arabidopsis CIB1 (cryptochrome-interacting basic-helix-loop-helix) protein is then indentified and characterized that interacts with CRY2 (cryptochrome 2) in a blue light–specific manner in yeast and Arabidopsis cells. Recently, a variety of microbial and plant-derived light-sensitive proteins have been engineered as optogenetic actuators, enabling high-precision spatiotemporal control of many cellular functions. So was the CRY2-CIB1 system, recently and successfully applied into mammalian cells in both vitro as well vivo.
3.Yeast-two-hybrid
Two-hybrid or interaction trap systems exploit the fact that transcription factors are comprised of two domains, a DNA binding domain (DBD) and an activation domain (AD). Two separate hybrid proteins are constructed in two-hybrid screens. The first hybrid protein is the DBD/protein X, while the second hybrid protein is the AD/protein Y. These two hybrids are encoded on separate yeast expression plasmids, with independent selectable markers. Based on the principle of yeast-two-hybrid, there are several pairs of plasmids designed for the screening of interacting protein library, commercially accessible.
4.Our project
Yeast-two-hybrid, the original method to discover the interaction between CRY2-CIB1, is now utilized in our project to realize spatiotemporal control of the target gene expression. Fusion proteins on one engineered plasmid, CRY2-GAL4 BD and CIB1-GAL4 AD are constitutively expressed. GAL4 BD is the domain of transcriptional factor GAL4, in charge of the binding to UAS in the promoter. Interaction between CRY2-CIB1 takes place under blue light, and GAL4 AD functions the expression of downstream CASP3, apoptosis is therefore happen[fig.7]

Fig.7 Iron sensitive aborbing system
Reference
In the mining industry, biomining, with its specific characteristics in energy conservation and emission reduction, is under accelerating development. Microbial consortium used in biomining mainly play the role as iron-oxidizer, sulfur-oxidizer or heterotrophic one. Microorganisms are also reported to be applied in the biomining’s following steps, like the metal recovery from the abandoned ore or slags, and Acid Mine Drainage(AMD) remediation. Fungi are the natural residents of AMD, with the ability of ferric iron oxidation and absorption, can take part into the biomining process and the following metal reduction operation. With the genetically engineering mediation, we may create a new colony of Saccharomyces cerevisiae with intensified potency in industrial practicing. For the biosafety reason, we would attach apoptosis system to ensure the engineering fungus shall not raise biological contamination. Due to the inefficiency in transformaiton and genetic manipulation, the “top down” method are widely adopted in the existing practice, which mesns indigenous microflora are inculated into the reactor and compete with each other. As far we know, It is the fisrt time introduing synthetic biology in biomining optimation in the logic of “bottom up”. The mature genetic manipulation in Saccharomyces cerevisiae ensures the feasibility, and perhaps our biobrick can be used in other organisms in the future, once engineering become facile for the other organisms.
Our microbial miner briefly consists of two systems, iron sensitive absorbing system and optogenetic apoptosis system.
II.Iron sensitive aborbing system
1.FET3 protein
High affinity iron uptake in Saccharomyces cerevisiae requires FET3 protein. Localized on the external cell surface, FET3 protein is proposed to facilitate iron uptake by catalyzing the oxidation of Fe(II) to Fe(III) by O2, shown as below:
4Fe(II) + O2 + 4H+ → 4Fe(III) + 2H2O
The posttranslational insertion of four copper ions into FET3 protein is essential for its activity, thus linking copper and iron homeostasis[fig.1]. In this model, Fe(III) is the substrate for the iron permease, encoded by FTR1 protein[fig2].

Fig.1 Ribbon diagram of FET3 protein. The T1, T2, and T3 copper atoms are shown in blue, green, and yellow, respectively. The three plastocyanin-like domains are shown as blue, green, and gold. Included in red are the carbohydrates resolvable in the electron density map.

Fig.2 FET3 and FTR1 proteins assemble into a complex in a cellular compartment early in the secretory pathway, most likely in the ER. The complex progresses to a post-Golgi compartment, where the CCC2 protein mediates copper delivery to FET3 protein. Finally, the copper-loaded F ET3 protein, presumably still complexed to FTR1 protein, is delivered to the plasma membrane and becomes competent for iron transport.
2.IRE-IRP1
Saccharomyces cerevisiae post-translationally regulates the expression of the plasma membrane high affinity iron transport system, consisting FET3 protein and FTR1 protein, in response to iron need. Incubation of cells in high iron leads to the internalization and degradation of both FET3 protein and FTR1 protein. Since what we expect is a way to accumulate iron that shall promote the absorption in presence of it, we cast our eyes to other mammalian regulation system involving iron.
In human cell, the cellular levels of ferritin and transferrin receptor (TfR) are also primarily regulated at translational level by changes in iron availability, through interactions between iron regulatory proteins (IRP1 and IRP2) and iron responsive elements (IRE) contained within the 5’ UTR of H- and L-ferritin mRNA and the 3’ UTR of TfR mRNA. When intracellular iron levels rise, IRP1 is no longer able to bind IRE, IRP2 is degraded and ferritin mRNA is efficiently translated. [fig.3] Apparently, the FET-IRP1 connetion from ferritin suits our anticipation to open the expression of objective gene. When iron level is high, IRP1 exist as a cytosolic aconitase. When iron islow, it assumes an open configuration associated with the loss of iron atoms in the iron-sulfur cluster, and can bind the IRE stem loop.[fig.4]

Fig.3 Decreased iron supply activates binding of IRPs to IRE resulting in translational inhibition of the mRNAs encoding ferritin and stabilization of the Tf R mRNA. During high iron conditions, IRPs lose their affinity for IREs, increasing translation of ferritin mRNAs and degradation of the Tf R mRNA.

Fig.4 The Fe-S cluster in aconitase.
3.Our project
What we plan is to integrate the FET3 from Saccharomyces cerevisiae with IRE-IRP1 system from human into the fungus, endowing it the ability of iron-sensitively enhanced aborption system. In the circumstance with low iron concentration, the constitutively expressing IRP1 bonds to the stem loop of IRE preceding the FET3 gene, impeding the expression of FET3. Once iron reaches to a high level, the breakdown of IRE-IRP1 connection shall activate the FET3 gene, which lead to more iron uptake, at last achieve the goal for iron accumulation.[fig.5]

Fig.5 Iron sensitive aborbing system
III.Optogenetic apoptosis system
1.Caspase-3
Caspase-3, a member of the caspase family of cell death proteases, cleaves cytoplasmic and nuclear substrates and promotes apoptotic cell death in mammalian cells[fig.6].

Fig.6 Extrinsic and intrinsic pathways to caspase-3 activation.
Although yeasts lack some elements of the complex apoptotic machinery of metazoan cells, recent studies show that many features of apoptosis, including a caspase-like activity, can be induced in these organisms by DNA damage and other apoptotic triggers. Here we plan to introduce CASP3 gene from human, and attempt to verify our hypothesis of the apoptosis effect in yeast by Caspase-3.
2.CRY2-CIB1
Cryptochromes (CRY) are photolyase-like blue-light receptors that mediate light responses in plants and animals. Arabidopsis CIB1 (cryptochrome-interacting basic-helix-loop-helix) protein is then indentified and characterized that interacts with CRY2 (cryptochrome 2) in a blue light–specific manner in yeast and Arabidopsis cells. Recently, a variety of microbial and plant-derived light-sensitive proteins have been engineered as optogenetic actuators, enabling high-precision spatiotemporal control of many cellular functions. So was the CRY2-CIB1 system, recently and successfully applied into mammalian cells in both vitro as well vivo.
3.Yeast-two-hybrid
Two-hybrid or interaction trap systems exploit the fact that transcription factors are comprised of two domains, a DNA binding domain (DBD) and an activation domain (AD). Two separate hybrid proteins are constructed in two-hybrid screens. The first hybrid protein is the DBD/protein X, while the second hybrid protein is the AD/protein Y. These two hybrids are encoded on separate yeast expression plasmids, with independent selectable markers. Based on the principle of yeast-two-hybrid, there are several pairs of plasmids designed for the screening of interacting protein library, commercially accessible.
4.Our project
Yeast-two-hybrid, the original method to discover the interaction between CRY2-CIB1, is now utilized in our project to realize spatiotemporal control of the target gene expression. Fusion proteins on one engineered plasmid, CRY2-GAL4 BD and CIB1-GAL4 AD are constitutively expressed. GAL4 BD is the domain of transcriptional factor GAL4, in charge of the binding to UAS in the promoter. Interaction between CRY2-CIB1 takes place under blue light, and GAL4 AD functions the expression of downstream CASP3, apoptosis is therefore happen[fig.7]

Fig.7 Iron sensitive aborbing system
Reference
- Spectral and Kinetic Properties of the Fet3 Protein from Saccharomyces cerevisiae, a Multinuclear Copper Ferroxidase Enzyme*. Richard F. Hassett‡, Daniel S. Yuan§¶, and Daniel J. Kosman‡i. THE JOURNAL OF BIOLOGICAL CHEMISTRY. Vol. 273, No. 36, Issue of September 4, pp. 23274–23282, 1998
- The Fet3 Protein: A Multicopper Ferroxidase Essential to Iron Metabolism in Yeast. Daniel J. Kosman. Handbook of Copper Pharmacology and Toxicology. March 1, 2002
- The copper-iron connection in biology: Structure of the metallo-oxidase Fet3p. Alexander B. Taylor*, Christopher S. Stoj†, Lynn Ziegler†, Daniel J. Kosman†, and P. John Hart*‡. PNAS October 25, 2005 vol. 102 no. 43 15459–15464.
- A Permease-Oxidase Complex Involved in High-Affinity Iron Uptake in Yeast.
- Robert Stearman, Daniel S. Yuan, Yuko Yamaguchi-Iwai, Richard D. Klausner andAndrew Dancis. Science, New Series, Vol. 271, No. 5255 (Mar. 15, 1996)
- IRON METABOLISM IN AN IN VITRO MODEL OF CARDIAC ISCHEMIA: HYPOXIC INJURY AND PROTECTIVE STRATEGIES. Virginia Cozzolino. UNIVERSITA’ DEGLI STUDI DI NAPOLI
- Post-transcriptional Regulation of the Yeast High Affinity Iron Transport System. M. Rosa Felice, Ivana De Domenico, Liangtao, Li, Diane McVey Ward, Beatrix Bartok, Giovanni Musci and Jerry Kaplan. J. Biol. Chem. 2005, 280:22181-22190.
- Post-transcriptional regulation of gene expression in response to iron deficiency: co-ordinated metabolic reprogramming by yeast mRNA-binding proteins. Sandra V. Vergara and Dennis J. Thiele1. Biochemical Society Transactions (2008) Volume 36, part 5.
- Translational repression by the human iron-regulatory factor (IRF) in Saccharomyces cerevisiae. Carla C.Oliveira, Britta Goossen1, Nilson l.T.Zanchin, John E.G.McCarthy*, Matthias W.Hentze1 and Renata Stripecke1. Nucleic Acids Research, 1993, Vol. 21, No. 23
- Emerging roles of caspase-3 in apoptosis. Alan G. Porter*,1 and Reiner U. JaÈ nicke1. Cell Death and Differentiation (1999) 6, 99 ± 104.
- Caspase-dependent apoptosis in yeast. Cristina Mazzoni, Claudio Falcone. Biochimica et Biophysica Acta 1783 (2008) 1320–1327.
- Apoptosis in Yeasts. Martin Weinberger, Lakshmi Ramachandran and William C. Burhans. IUBMB Life, 55(8): 467–472, August 2003.
- A Caspase-Related Protease Regulates Apoptosis in Yeast. Frank Madeo,1,7 Eva Herker,1 Corinna Maldener,1
- Silke Wissing,1 Stephan La¨ chelt,1 Mark Herlan,2, Markus Fehr,3 Kirsten Lauber,4 Stephan J. Sigrist,5, Sebastian Wesselborg,4 and Kai-Uwe Fro¨ hlich6. Molecular Cell, Vol. 9, 911–917, April, 2002
- Photoexcited CRY2 Interacts with CIB1 to Regulate Transcription and Floral Initiation in Arabidopsis. Hongtao Liu, Xuhong Yu, Kunwu Li, John Klejnot, Hongyun Yang, Dominique Lisiero, Chentao Lin*. SCIENCE VOL 322 5 DECEMBER 2008.
- Optical control of mammalian endogenous
- transcription and epigenetic states. Silvana Konermann1,2*, Mark D. Brigham1,2,3*, Alexandro Trevino1,2, Patrick D. Hsu1,2,4, Matthias Heidenreich1,2, Le Cong1,2,5, Randall J. Platt1,2, David A. Scott1,2, George M. Church1,6 & Feng Zhang1,2. doi:10.1038/nature12466
Experiment
Preliminary construction of the Cry2-CIB1 system
I.Cloning of the Cry2 & CIB1 gene
According to the sequences from NCBI, we designed two pairs of primers respectively, and cloned them based on the cDNA library of Arabidopsis. To fulfill the requirement of following gateway operation, the attB1/2 are added to the primers.
Primer for Cry2:
Fwd 5’→3’ GTACAAAAAAGCAGATGAATGGAGCTATAGGA
attB1
Rev 5’→3’ GTACAAGAAAGCTGGGTTCAAACTCCTAAATTGCC
attB2
Primer for CIB1:
Fwd 5’→3’ GTACAAAAAAGCAGATGAAGATGGACAAAAAGA
attB1
Rev 5’→3’ GTACAAGAAAGCTGGGTTCATTTGCAACCATTTTT
attB1
II.Yeast-two-hybrid
What we chose is the commercial shuttle vectors, pDEST32 and pDEST22[fig.1], where the DBD and AD is actually Gal4 BD and Gal4 AD respectively, and the Gal4 AD shall specifically bind to the upstream activating sequence(UAS), locating in the promoters of several reporter genes in the yeast strain genome like lacZ, HIS3 as well URA3.
Fig.2 The map of our two plasmids: pDEST22 and pDEST32

Since we have obtained CRY2 and CIB1, what we do next is to induce both of them into their vectors with the technology named Gateway, constructing two new plasmids, pDEST22-CIB1 and pDEST32-32 as below[fig.2]. Then we transform the plasmids above into the Saccharomyces cerevisiae cell, AH109, screening by Leu and Trp auxotroph.
Fig.3 The map of our two plasmids: pDEST22-CIB1 and pDEST32-CRY2

III. Optogenetic test
To test the effect of light switchable gene expression system, blue-white selection is induced when the blue light in special incubator[fig.3] shall initiate the expression of LacZ, which combined with the IPTG and X-gal in solid medium, makes the colony into blue, contrarily, colony in dark remains white.[fig.4]
Fig.3 Blue light incubator

Fig.4 The positive result of yeast X-gal coloration

Cloning of the genes we need and ligated them into of T-vector
I.IRE-IRP
1.IRP
We decide to clone the IRP from human cDNA library.
Primers for IRP[fig.5]:
Fwd 5’→3’ GCGAAGCTTTCAGTAATCATGAGCAAC
HindIII
Rev 5’→3’ GCGGAGCTCTTGAGCAGAGCGTAAGA
SacI
Fig.5

2.IRE
Since IRE is a quite short sequence, counting 40 bp at all. A pair of single-stranded DNA are synthesized as the primers, and annealing shall create double-stranded DNA.
Pair of single-stranded DNAs
Fwd 5’→3’ GCGAAGCTTGTTCTTGCTTCAACAGTGTTTGGACGGAACACTAGTGCG
HindIII SpeI
Rev 5’→3’ CGCACTAGTGTTCCGTCCAAACACTGTTGAAGCAAGAACAAGCTTCGC
SpeI HindIII
II.FET3
1.Prepare for the genome of Saccharomyces cerevisiae
Due to the lack of existing yeast genome as the PCR template, we order a yeast genome extraction kit to prepare the source.[fig.2]
Fig.2
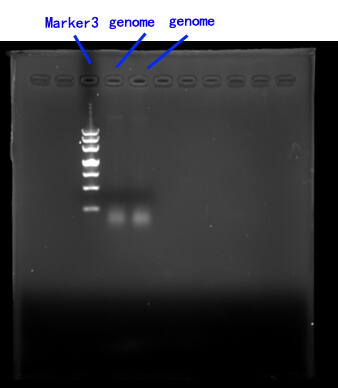
2.Cloning of FET3
Primers for FET3[fig.3]
Fwd 5’→3’ GCGAAGCTTATGACTAACGCTTTGCTCTCTATAG
HindIII
Rev 5’→3’ GCGGAGCTCTGGAACCCTTGACCGA
SacI
Fig.3
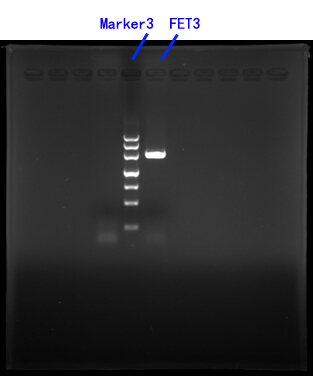
III.CASP3
Caspase-3 normally exists in the cytosolic fraction of cells as an inactive precursor that is activated proteolytically when cells are signaled to undergo apoptosis.[4] So in the project, we want to make the modified yeast commit suicide through overexpress caspase-3 protein to protect natural environment.
The template for PCR is also the human cDNA library.
Primers for CASP3[fig.4]
Fwd 5’→3’ GCGACTAGTGCTCTGGTTTTCGGTGGG
Spe1
Rev 5’→3’ GCGGAGCTCTGGAACCCTTGACCGA
SacI
Fig.4

IV.TA cloning
To make sure the PCR-cloned sequences are expected, we ligate them into the pMD18-T vector, then sequence all together. On the other hand, objective sequence in circular plasmids makes it convenient and stable for storage for quite long time.
Construction of the two plasmids in iron sensitive absorbing system
Since we comfronted trouble in the ligation of gene with shuttle vector by T4 ligase after restriction enzyme treatment, the schematics below is just our original plan.
I.pDEST22-IRP1
Concerning the limitation of restriction cloning site, We have to ligated IRP1 firstly with pDEST32, and finally tranfer it into the pDEST22 vector.
1.When pDEST32 are treated with HindIII SacI, it yields segment of 2.3K & 10K.
2.When T-IRP1 are treated with HindIII SacI, it yields segment of 2.6K.
3.We can ligate the 10K and 2.6K segments together, constructing pDEST32-IREBP1.
4.Restriction enzymens XhoI and SacI digest pDEST22 and pDEST32-IREBP1 respectively.
5.The pDEST22-IREBP1 comes from the ligation of 5.1K segment from pDEST22 yields and the 4.1K segment from pDEST32-IREBP1 yields.
II.pDEST32-IREBP1
1.Restriction enzymens HindIII and SacI digest pDEST32 and T-IRE respectively.
2.pDEST32-IRE is the ligation product of 10K segment from pDEST32 and IRE
3.Restriction enzymens SpeI and SacI digest pDEST32-IRE and T-FET3 respectively.
4.pDEST32-IRE-FET3 is the ligation product of 10K segment from pDEST32-IRE and 2K segment from T-FET3
Result
Gene cloning
The genes we have cloned:
1.IRP and CASP3 are from existing cDNA library of human.
2.CRY2, CIB1 are from existing cDNA library of Arabidopsis.
3.FET3 is from the genome of Saccharomyces cerevisiae made by ourselves.
4.IRE is made by annealing of two synthetic sigle-strain DNA.
TA cloning
For sequencing and the stability of conservation, IRP, CASP3, FET3 and IRE are all introduced into the pMD-18T vector.
Optogenetic yeast-two-hybrid
CRY2 and CIB1 are introduced into the commercial shuttle vectors, pDEST32 and pDEST22, by the gateway operation. Both of the pDEST22-CIB1 and pDEST32-CRY2 are transformed into the Saccharomyces cerevisiae cell AH109. Auxotroph screening and X-gal coloration prove the viability for the blue light to mediate CRY2-CIB1 interaction.
Future work
Step I.
When all of the schematic work finished, we are firstly going to verify the hypothesis of the apoptosis effect when caspase-3 expressed in yeast.
Step II.
The kinetic parameter of the light mediation, iron sensing and iron absorption are with worth to detect.
Step III.
Though we would like to see them in only one microorganism clone, according to the original design, iron sensitive absorbing system and optogenetic apoptosis system consists of two engineered plasmids repectively. Four plasmids in total makes it impossible for integration. Further genetically engineering design and operation are necessary, mainly in system simplification.
 "
"







