Team:Paris Saclay/Notebook/September/2
From 2014.igem.org
Fanny Boulet (Talk | contribs) |
m (→Labwork) |
||
| Line 1: | Line 1: | ||
{{Team:Paris_Saclay/notebook_header}} | {{Team:Paris_Saclay/notebook_header}} | ||
| + | =Tuesday 2nd September= | ||
| + | |||
==Labwork== | ==Labwork== | ||
| Line 131: | Line 133: | ||
====pPS5==== | ====pPS5==== | ||
To have more plasmid We do some bacteria culture (pPS5) | To have more plasmid We do some bacteria culture (pPS5) | ||
| + | |||
| + | ==Photo of the Day== | ||
| + | [[File:Paris Saclay 2_september.jpg|600px|center]] | ||
{{Team:Paris_Saclay/notebook_footer}} | {{Team:Paris_Saclay/notebook_footer}} | ||
Revision as of 00:09, 14 October 2014
Contents |
Tuesday 2nd September
Labwork
B-Contruction of the fusion protein
By Hv
We can see that the blue colonies of E.coli are not blue in a simple petri dish with ampicillin. It's a fail. So, to figure our problem we decided to do a sequencing of our plasmids.
We chose 6 colonies from an Xgal-IPTG petri dish:
- 1 completely blue
- 2 blue with a white ring around the blue
- 3 white colony
PS: we found that there is 2 types of blue colonies: completely blue and blue with a white ring around the blue)
We first did a liquid culture in 5 mL of LB.
D- Lemon scent
By Melanie
Limonene synthase
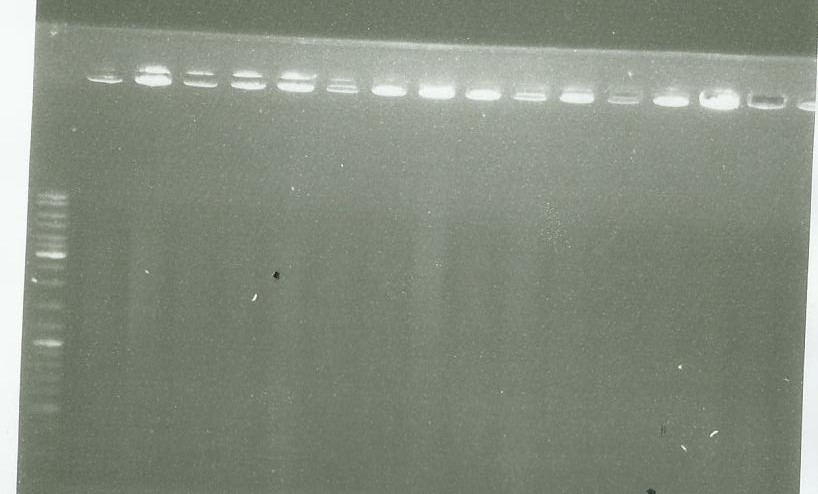
Migration of the PCR done yesterday
picture 1 well 1= ladder well 2-6 = pPS5 other well = LS PCR
picture 2 = LS PCR As we see, there are nothing on our gel so we conclude that we failed to clone LS in pGMETeasy
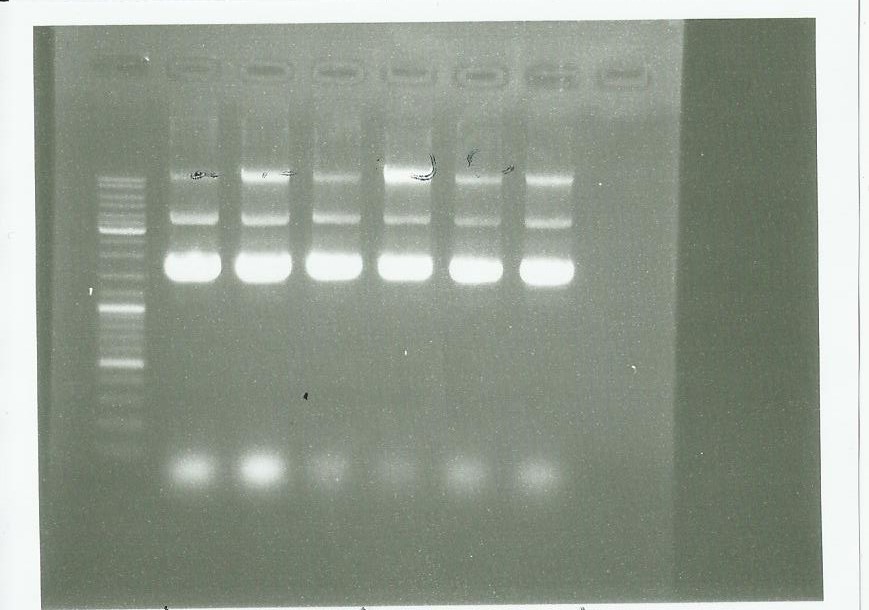
pPPS5
the photo in the same as previously: We saw a band but we will check another time by PCR to be sure.
pPS3 and pPS4
Plasmid extraction using the plasmid DNA extraction kit and electrophoresis:
We conclude that we have done a good manipulation.
Limonene synthase PCR
As we saw that we don't have anything for LS, We try to do other PCR from the Biobrick (BBA762100). We use two enzyme: Dream taq and Vent taq
| component | volume |
|---|---|
| H2O | 42μl |
| buffer | 5μl |
| dNTPs | 1μl |
| iPS66 | 1μl |
| iPS67 | 1μl |
| DNA | 0.5μl |
| enzyme | 0.5μl |
Tubes were placed in PCR machine with the following parameters.
| Cycle step | Temperature | Time | Cycle |
|---|---|---|---|
|
Initial denaturation |
94°C |
1 min |
1 |
| Denaturation | 94°C | 30 s | 25 |
| Annealing | 50°C | 25 s | 25 |
| Extension | 72°C | 1 min | 25 |
| Final extension | 72°C | 10 min | 1 |
| Final extension | 8°C | hold | 1 |
pPS5
To have more plasmid We do some bacteria culture (pPS5)
 "
"