Before we get started:
Biofilm formation is part of the reason S. mutans is so devastating to oral health (see [Project Overview] for more information). For adequate oral protection, biofilm formation is a problem we must solve, and we can do so internally, by modifying the S. mutans genome, (see [Control: Inhibitor]), or externally, via a modified probiotic. This means we need to know about conditions unique to biofilms in order to design a sensor.
What happens to our teeth if it is covered in biofilm? Metabolic acids which are waste products of S. mutans naturally begin to accumulate to very high concentrations in the local space, and cannot disperse out into the rest of the mouth. This results in a low pH in areas covered by biofilms, about 4.0~5.0. Since our probiotic is physically close to S. mutans (see [Cleanse: Attachment]), if our probiotic detects a low pH, there’s a good chance it is within a biofilm. Figure 1 depicts this in a simplified diagram.

Figure 1.
Now that we know the target pH range, we need to find out what biofilms are made of, and how to get rid of them. Biofilms are actually conglomerated bacteria colonies enmeshed in a self-synthesized layer of extracellular polysaccharide matrix. Of these, PGA, or poly-gamma glutamate, has been demonstrated to be a main component across the biofilm of several phylogenetically diverse bacterial species, and polyglycine bridges can cross-link glycopeptides that make up the cell walls of biofilm-producing bacteria.
The ideal biofilm degrader must be able to target some, if not all, of these elements.
So how did we do it?
After careful literature search, we found two promising enzymes:
Lysostaphin is a zinc-containing enzyme 27kDa in size, taken from the genome of Staphylococcus simulans, and was used previously by the [2012 HIT-Harbin iGEM team (TO ADD!)]. It is particularly powerful because it combines the actions of three different peptidoglycan degradation enzymes, and as such can target a wide variety of the glycopeptides that form cell walls, including features such as the polyglycine bridge. Also, unlike other enzymes of this type, it can target both actively growing and non-dividing cells. To add to its merits, lysostaphin causes no side effects in humans.
Dispersin B comes from Aggregatibacter actinomycetemcomitans, a Gram-negative rod oral bacteria. It is a soluble glycoside hydrolase capable of degrading PGA.
After the enzymes have been decided, we needed to find a promoter responsive to the conditions within a biofilm. The Asr promoter comes from E. coli strain MG1655, and is induced by extracellular low pH of between 4.0~5.0. Perfect for what we need.
But wait! We’re not quite done yet. Even after being equipped with the Asr promoter, lysostaphin, and dispersin B, our probiotic would still be useless against biofilms unless we export the enzymes out of the cell, where they can do their jobs. Here’s where the signal protein comes in.
YebF is a signal protein naturally secreted by E. coli, though experimental strains rarely secrete this protein, which makes it useful in synthetic biology. Proteins tacked onto YebF can be expected to also be secreted outside of the host cell, so we put this in front of our enzymes.
At last, our circuit is complete!

Our expectation is that the three-protein fusion of YebF-Lysostaphin-Dispersin B will be exported out of the cell upon translation by the YebF, where they will be able to do their job of degrading the biofilm, such as in Figure 2.
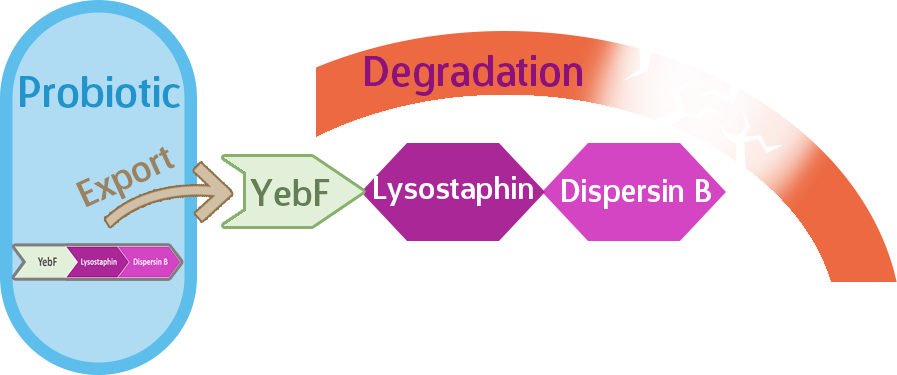
Figure 2.
Putting it to the test!
Having a circuit is only the start! Now we must test how well our circuit functions. For the Antibiofilm circuit, we need to test specifically for three things: the function of the Asr promoter, the function of YebF, and the function of lysostaphin and dispersin B.
Testing the Function of the Asr Promoter
For this test we constructed the following circuit in E. coli. We aim to elucidate the exact pH-activity relation by subjecting the circuit to conditions of various pH, and measuring activity via RFP fluorescence intensity, quantized in OD.

Here is the details of our method:
- Transform the circuit to E. coli, and grow the modified E.coli on agar plates overnight.
- Select the strain that expresses growth differences from the previous step. Prepare two of each liquid culture tubes for pH values 3.5, 4, 4.5, 5, 5.5, 6, 6.5, and 7, and grow the selected strain in all tubes. Use the growth data from each tube to plot the precise effect of pH on the Asr promoter.
- Measure the OD value (RFP/bacterium number), and plot out curves.
Testing the Function of YebF
Since we also used YebF in [Control: Target], we have done our functional assays there. Feel free to check it out if you’re interested!
Testing the Function of Lysostaphin and Dispersin B
For this part of the test, we designed a novel apparatus we called the [HOPE Box (TO ADD)], which allowed us to culture S. mutans in real saliva (yes, real saliva!) and observe their biofilm formation. The HOPE Box provided us with the most accurate representation of the human mouth that we could obtain!
Our result
Reference
- Na, D., Yoo, S. M., Chung, H., Park, H., Park, J. H., & Lee, S. Y. (January 01, 2013). Metabolic engineering of Escherichia coli using synthetic small regulatory RNAs. Nature Biotechnology, 31, 2, 170-4.
- Li, Y.-H., Lau, P. C. Y., Tang, N., Svensater, G., Ellen, R. P., & Cvitkovitch, D. G. (November 15, 2002). Novel Two-Component Regulatory System Involved in Biofilm Formation and Acid Resistance in Streptococcus mutans. Journal of Bacteriology, 184, 22, 6333-6342.
- Baev, D., England, R., & Kuramitsu, H. K. (January 01, 1999). Stress-induced membrane association of the Streptococcus mutans GTP-binding protein, an essential G protein, and investigation of its physiological role by utilizing an antisense RNA strategy. Infection and Immunity, 67, 9, 4510-6.
- Yoshida, A., & Kuramitsu, H. K. (December 01, 2002). Multiple Streptococcus mutans Genes Are Involved in Biofilm Formation. Applied and Environmental Microbiology, 68, 12, 6283-6291.
- Biswas, I., Jha, J. K., & Fromm, N. (August 01, 2008). Shuttle expression plasmids for genetic studies in Streptococcus mutans. Microbiology, 154, 8, 2275-2282.
- Li, Yung-Hua, Tang, Nan, Aspiras, Marcelo B., Lau, Peter C. Y., Lee, Janet H., Ellen, Richard P., & Cvitkovitch, Dennis G. (n.d.). A Quorum-Sensing Signaling System Essential for Genetic Competence in Streptococcus mutans Is Involved in Biofilm Formation. American Society for Microbiology.
- Wu, J., Cho, M. I., & Kuramitsu, H. K. (January 01, 1995). Expression, purification, and characterization of a novel G protein, SGP, from Streptococcus mutans. Infection and Immunity, 63, 7, 2516-21.
 "
"