Before we get started:
The "control part" is going to control the number of S. mutans. When the number of S. mutans exceeds the threshold that causes cavities, the circuit will be activated, thus killing the excess S. mutans.
In order to find and confirm the threshold that causes cavities, we designed different kinds and numbers of terminators (each with different leakage rates) to create the threshold. Moreover, we combined both wet lab results and modelling to decide which design is more suitable.
Over the years, it has been found that the competence stimulating peptide, CSP, a quorum sensing chemical, is released in every competence S. mutans. Thus, we can detect the number of S. mutans by marking the amount of CSP.
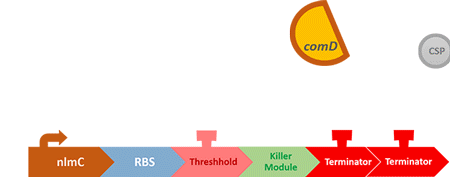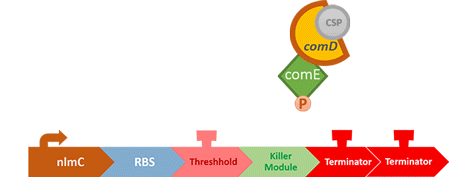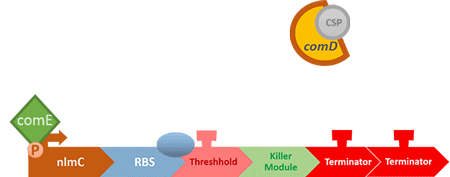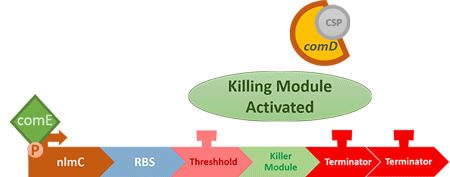
In S. mutans the CSP bind to the membrane receptor, "comD", thereby phosphorylating the response regulator, "comE". The phosphorylated comE will activate numerous vital biological mechanisms in Streptococcus mutans such as biofilm formation and the release of mutacin, an antibiotic peptide. Compared with all the other mechanisms involved with CSP, it is found that the promoter of gene "nlmC"( non-lantibiotic mutacin C) in S. mutans has the best performance under the stimulation of CSP.
At this point, the two main organisms we adapted for our project are our probiotics and M102 bacteriophage. We used one of the most commonly seen quorum sensing chemicals, AHL, N-Acyl-homoserine-lactone, as our indicator between phage and E.coli. Due to the idea of phage threshold control of the number of S. mutans in prevention of the second-dominant strain in the oral cavity, we envision a possibility to trigger another killing system to reach synergistic effect on diminishing S. mutans. If the threshold terminators were passed, the downstream signaling pathway would be switched on and contact with the engineered probiotics.
When the amount of the S. mutans is so large that the phages couldn't afford the loading, CSP would pass the threshold value and turn on the luxR-luxI system. LuxI acts as a gene which would generate AHL-synthase that yielding the quorum-sensing signal AHL right after the threshold level of S. mutans is exceeded. LuxR is a protein which forms a complex with AHL, and then turns into an activated form which will afterwards bind with the promoter pLuxR and trigger it. The signal sequence and endolysin downstream would be secreted and eliminate the excess S. mutans.
So how did we do it?
Putting it to the test!
Our result
Reference
- http://www.expertsmind.com/topic/microbiology/bacterial-cell-wall-92313.aspx
 "
"