Team:Aachen/Project/FRET Reporter
From 2014.igem.org
(→A Fluorescence Answer Faster than Expression) |
m (→Comparing the kinetic of the double plasmid systems K1319013 + K1319008 and K1319014 + K1319008 with standard GFP expression) |
||
| (19 intermediate revisions not shown) | |||
| Line 15: | Line 15: | ||
<li style="width: 156px;margin-left: 8px;margin-right: 8px;"> | <li style="width: 156px;margin-left: 8px;margin-right: 8px;"> | ||
<a class="menulink" href="https://2014.igem.org/Team:Aachen/Project/FRET_Reporter#fluorescence" style="color:black"> | <a class="menulink" href="https://2014.igem.org/Team:Aachen/Project/FRET_Reporter#fluorescence" style="color:black"> | ||
| - | <div class="menusmall-item menusmall-info" ><div class="menukachel" style="line-height: 1.5em | + | <div class="menusmall-item menusmall-info" ><div class="menukachel" style="top:32%; line-height:1.5em;">A Faster Answer</div></div> |
<div class="menusmall-item menusmall-img" style="background: url(https://static.igem.org/mediawiki/2014/0/0b/Aachen_14-10-13_GFP_iNB.png); norepeat scroll 0% 0% transparent; background-size:100%"> | <div class="menusmall-item menusmall-img" style="background: url(https://static.igem.org/mediawiki/2014/0/0b/Aachen_14-10-13_GFP_iNB.png); norepeat scroll 0% 0% transparent; background-size:100%"> | ||
</div> | </div> | ||
| Line 23: | Line 23: | ||
<li style="width: 156px;margin-left: 8px;margin-right: 8px;"> | <li style="width: 156px;margin-left: 8px;margin-right: 8px;"> | ||
<a class="menulink" href="https://2014.igem.org/Team:Aachen/Project/FRET_Reporter#fret" style="color:black"> | <a class="menulink" href="https://2014.igem.org/Team:Aachen/Project/FRET_Reporter#fret" style="color:black"> | ||
| - | <div class="menusmall-item menusmall-info" ><div class="menukachel" style="line-height: 1.5em | + | <div class="menusmall-item menusmall-info" ><div class="menukachel" style="top:32%; line-height:1.5em;">The FRET System</div></div> |
<div class="menusmall-item menusmall-img" style="background: url(https://static.igem.org/mediawiki/2014/5/54/Aachen_14-10-13_FRET_Arrows_iNB.png); norepeat scroll 0% 0% transparent; background-size:100%"> | <div class="menusmall-item menusmall-img" style="background: url(https://static.igem.org/mediawiki/2014/5/54/Aachen_14-10-13_FRET_Arrows_iNB.png); norepeat scroll 0% 0% transparent; background-size:100%"> | ||
</div> | </div> | ||
| Line 31: | Line 31: | ||
<li style="width: 156px;margin-left: 8px;margin-right: 8px;"> | <li style="width: 156px;margin-left: 8px;margin-right: 8px;"> | ||
<a class="menulink" href="https://2014.igem.org/Team:Aachen/Project/FRET_Reporter#darkquencher" style="color:black"> | <a class="menulink" href="https://2014.igem.org/Team:Aachen/Project/FRET_Reporter#darkquencher" style="color:black"> | ||
| - | <div class="menusmall-item menusmall-info" ><div class="menukachel" style="line-height: 1.5em | + | <div class="menusmall-item menusmall-info" ><div class="menukachel" style="top:32%; line-height:1.5em;">REACh Quenchers</div></div> |
<div class="menusmall-item menusmall-img" style="background: url(https://static.igem.org/mediawiki/2014/6/65/Aachen_14-10-13_REACh_iNB.png); norepeat scroll 0% 0% transparent; background-size:100%"> | <div class="menusmall-item menusmall-img" style="background: url(https://static.igem.org/mediawiki/2014/6/65/Aachen_14-10-13_REACh_iNB.png); norepeat scroll 0% 0% transparent; background-size:100%"> | ||
</div> | </div> | ||
| Line 39: | Line 39: | ||
<li style="width: 156px;margin-left: 8px;margin-right: 8px;"> | <li style="width: 156px;margin-left: 8px;margin-right: 8px;"> | ||
<a class="menulink" href="https://2014.igem.org/Team:Aachen/Project/FRET_Reporter#gfp-reach" style="color:black"> | <a class="menulink" href="https://2014.igem.org/Team:Aachen/Project/FRET_Reporter#gfp-reach" style="color:black"> | ||
| - | <div class="menusmall-item menusmall-info" ><div class="menukachel" style="line-height: 1.5em | + | <div class="menusmall-item menusmall-info" ><div class="menukachel" style="top:32%; line-height:1.5em;">The Fusion Protein</div></div> |
<div class="menusmall-item menusmall-img" style="background: url(https://static.igem.org/mediawiki/2014/0/02/Aachen_14-10-13_Fusion_Protein_iNB.png); norepeat scroll 0% 0% transparent; background-size:100%"> | <div class="menusmall-item menusmall-img" style="background: url(https://static.igem.org/mediawiki/2014/0/02/Aachen_14-10-13_Fusion_Protein_iNB.png); norepeat scroll 0% 0% transparent; background-size:100%"> | ||
</div> | </div> | ||
| Line 54: | Line 54: | ||
<li style="width: 156px;margin-left: 8px;margin-right: 8px;"> | <li style="width: 156px;margin-left: 8px;margin-right: 8px;"> | ||
<a class="menulink" href="https://2014.igem.org/Team:Aachen/Project/FRET_Reporter#tevprotease" style="color:black"> | <a class="menulink" href="https://2014.igem.org/Team:Aachen/Project/FRET_Reporter#tevprotease" style="color:black"> | ||
| - | <div class="menusmall-item menusmall-info" ><div class="menukachel" style="line-height: 1.5em | + | <div class="menusmall-item menusmall-info" ><div class="menukachel" style="top:32%; line-height:1.5em;">TEV Protease</div></div> |
<div class="menusmall-item menusmall-img" style="background: url(https://static.igem.org/mediawiki/2014/d/dc/Aachen_14-10-13_TEV_Protease_iNB.png); norepeat scroll 0% 0% transparent; background-size:100%"> | <div class="menusmall-item menusmall-img" style="background: url(https://static.igem.org/mediawiki/2014/d/dc/Aachen_14-10-13_TEV_Protease_iNB.png); norepeat scroll 0% 0% transparent; background-size:100%"> | ||
</div> | </div> | ||
| Line 62: | Line 62: | ||
<li style="width: 156px;margin-left: 8px;margin-right: 8px;"> | <li style="width: 156px;margin-left: 8px;margin-right: 8px;"> | ||
<a class="menulink" href="https://2014.igem.org/Team:Aachen/Project/FRET_Reporter#reachachievements" style="color:black"> | <a class="menulink" href="https://2014.igem.org/Team:Aachen/Project/FRET_Reporter#reachachievements" style="color:black"> | ||
| - | <div class="menusmall-item menusmall-info" ><div class="menukachel" style="line-height: 1.5em | + | <div class="menusmall-item menusmall-info" ><div class="menukachel" style="top:32%; line-height:1.5em;">Achieve-<br/>ments</div></div> |
<div class="menusmall-item menusmall-img" style="background: url(https://static.igem.org/mediawiki/2014/e/ef/Aachen_14-10-15_Medal_Cellocks_iNB.png); norepeat scroll 0% 0% transparent; background-size:100%"> | <div class="menusmall-item menusmall-img" style="background: url(https://static.igem.org/mediawiki/2014/e/ef/Aachen_14-10-15_Medal_Cellocks_iNB.png); norepeat scroll 0% 0% transparent; background-size:100%"> | ||
</div> | </div> | ||
| Line 70: | Line 70: | ||
<li style="width: 156px;margin-left: 8px;margin-right: 8px;"> | <li style="width: 156px;margin-left: 8px;margin-right: 8px;"> | ||
<a class="menulink" href="https://2014.igem.org/Team:Aachen/Project/FRET_Reporter#reachoutlook" style="color:black"> | <a class="menulink" href="https://2014.igem.org/Team:Aachen/Project/FRET_Reporter#reachoutlook" style="color:black"> | ||
| - | <div class="menusmall-item menusmall-info" ><div class="menukachel" style=" | + | <div class="menusmall-item menusmall-info" ><div class="menukachel" style="top:40%;">Outlook</div></div> |
<div class="menusmall-item menusmall-img" style="background: url(https://static.igem.org/mediawiki/2014/6/67/Aachen_14-10-16_Outlook_Cellocks_iNB.png); norepeat scroll 0% 0% transparent; background-size:100%"> | <div class="menusmall-item menusmall-img" style="background: url(https://static.igem.org/mediawiki/2014/6/67/Aachen_14-10-16_Outlook_Cellocks_iNB.png); norepeat scroll 0% 0% transparent; background-size:100%"> | ||
</div> | </div> | ||
| Line 102: | Line 102: | ||
'''Advantages of the novel biosensor:''' | '''Advantages of the novel biosensor:''' | ||
| - | * When ''P. aeruginosa'' is detected by our cells, the reporter protein has already been expressed and only waits to be activated. The cleavage reaction catalyzed by the TEV protease is a faster process than expression and correct folding of GFP. Therefore, our sensor cells provide an exceptionally'''early response'''. | + | * When ''P. aeruginosa'' is detected by our cells, the reporter protein has already been expressed and only waits to be activated. The cleavage reaction catalyzed by the TEV protease is a faster process than expression and correct folding of GFP. Therefore, our sensor cells provide an exceptionally '''early response'''. |
| - | * While a certain concentration of homoserine lactone will produce the same number of gene read-outs and the TEV protease is about the same size as GFP (~27 kDa), one TEV protease can cleave many GFP-REACh constructs per time than fluorescent protein molecules could be expressed and go through fluorescence maturation. Through this additional reaction, we introduce an '''amplification step''' into our system. Hypothetically, the TEV protease will enable the production of '''a much stronger signal''' in a | + | * While a certain concentration of homoserine lactone will produce the same number of gene read-outs and the TEV protease is about the same size as GFP (~27 kDa), one TEV protease can cleave many GFP-REACh constructs per time than fluorescent protein molecules could be expressed and go through fluorescence maturation. Through this additional reaction, we introduce an '''amplification step''' into our system. Hypothetically, the TEV protease will enable the production of '''a much stronger signal''' in a given time interval. |
In parallel, we worked on a [https://2014.igem.org/Team:Aachen/Project/Model model] of the respective biosensor system, in order to confirm our hypothesis of a faster and stronger fluorescence response than a conventional biosensor would povide. | In parallel, we worked on a [https://2014.igem.org/Team:Aachen/Project/Model model] of the respective biosensor system, in order to confirm our hypothesis of a faster and stronger fluorescence response than a conventional biosensor would povide. | ||
| Line 118: | Line 118: | ||
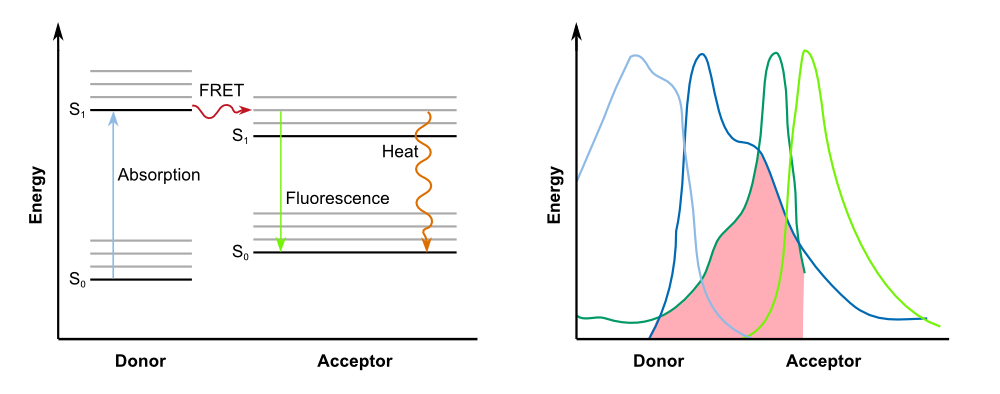
Förster resonance energy transfer (FRET), sometimes also called fluorescence resonance energy transfer, is a physical process of energy transfer. In FRET, the energy of a donor chromophore, whose electrons are in an excited state, is passed to a second chromophore, the acceptor. The '''energy is transferred without radiation''' and is therefore not exchanged via emission and absorption of photons. The acceptor then releases the energy received from the donor, for example, as light of a longer wavelength. | Förster resonance energy transfer (FRET), sometimes also called fluorescence resonance energy transfer, is a physical process of energy transfer. In FRET, the energy of a donor chromophore, whose electrons are in an excited state, is passed to a second chromophore, the acceptor. The '''energy is transferred without radiation''' and is therefore not exchanged via emission and absorption of photons. The acceptor then releases the energy received from the donor, for example, as light of a longer wavelength. | ||
| - | In biochemistry and cell biology | + | In biochemistry and cell biology fluorescent dyes, which interact via FRET, are applied as "optical nano metering rules", because the intensity of the transfer is dependent on the spacing between donor and acceptor. FRET can be observed over distances of up to 10 nm. This way, protein-protein interactions and conformational changes of a variety of tagged biomolecules can be observed. For an efficient FRET to occur, there must be a substantial overlap between the donor fluorescence emission spectrum and the acceptor fluorescence excitation (or absorption) spectrum ([http://www.nature.com/nprot/journal/v8/n2/full/nprot.2012.147.html Broussard et al., 2013]), as described in the figure below. |
{{Team:Aachen/Figure|Aachen_14-10-07_Jablonski_Diagram_and_Absorption_Spectra_iNB.png|align=center|title=FRET betweeen donor and acceptor|subtitle=On the left: Jablonski diagram showing the transfer of energy between donor and acceptor; on the right: For successful FRET, the emission spectrum of of the donor has to overlap with the absorption spectrum of the acceptor.|width=900px}} | {{Team:Aachen/Figure|Aachen_14-10-07_Jablonski_Diagram_and_Absorption_Spectra_iNB.png|align=center|title=FRET betweeen donor and acceptor|subtitle=On the left: Jablonski diagram showing the transfer of energy between donor and acceptor; on the right: For successful FRET, the emission spectrum of of the donor has to overlap with the absorption spectrum of the acceptor.|width=900px}} | ||
| - | However, '''fluorescence is not an essential requirement for FRET'''. This type of energy transfer can also be observed between donors that are capable of other forms of radiation, such as phosphorescence, bioluminescence or chemiluminescence | + | However, '''fluorescence is not an essential requirement for FRET'''. This type of energy transfer can also be observed between donors that are capable of other forms of radiation, such as phosphorescence, bioluminescence or chemiluminescence. Acceptor chromophores do not necessarily emit the energy in form of light, and can lead to quenching instead. Thus, this kind of acceptors are also referred to as '''dark quenchers'''. In our project, we use a FRET system with a dark quencher, namely our '''REACh construct'''. |
| Line 136: | Line 136: | ||
</center> | </center> | ||
| - | In 2006, [http://www.pnas.org/content/103/11/4089.full Ganesan et al.] were the first to present a previously undescribed FRET acceptor, a non-fluorescent yellow fluorescent protein (YFP) mutant called '''REACh ( | + | In 2006, [http://www.pnas.org/content/103/11/4089.full Ganesan et al.] were the first to present a previously undescribed FRET acceptor, a non-fluorescent yellow fluorescent protein (YFP) mutant called '''REACh (Resonance Energy-Accepting Chromoprotein)'''. YFP can be used as a FRET acceptor in combination with GFP as the donor in FRET microscopy and miscellaneous assays in molecular biology. The ideal FRET couple should possess a large spectral overlap between donor emission and acceptor absorption - as illustrated in previous section - but have separated emission spectra to allow their selective imaging. |
To optimize the spectral overlap of this FRET pair, the group obtained '''a genetically modified YFP acceptor'''. Mutations of amino acid residues that stabilize the excited state of the chromophore in enhanced YFP (EYFP) resulted in a non-fluorescent chromoprotein. Two mutations, H148V and Y145W, reduced the fluorescence emission by 82 and 98%, respectively. Ganesan et al. chose the Y145W mutant and the Y145W/H148V double mutant as FRET acceptors, and named them REACh1 and REACh2, respectively. '''Both REACh1 and REACh2 act as dark quenchers of GFP'''. | To optimize the spectral overlap of this FRET pair, the group obtained '''a genetically modified YFP acceptor'''. Mutations of amino acid residues that stabilize the excited state of the chromophore in enhanced YFP (EYFP) resulted in a non-fluorescent chromoprotein. Two mutations, H148V and Y145W, reduced the fluorescence emission by 82 and 98%, respectively. Ganesan et al. chose the Y145W mutant and the Y145W/H148V double mutant as FRET acceptors, and named them REACh1 and REACh2, respectively. '''Both REACh1 and REACh2 act as dark quenchers of GFP'''. | ||
| Line 148: | Line 148: | ||
<span class="anchor" id="gfp-reach"></span> | <span class="anchor" id="gfp-reach"></span> | ||
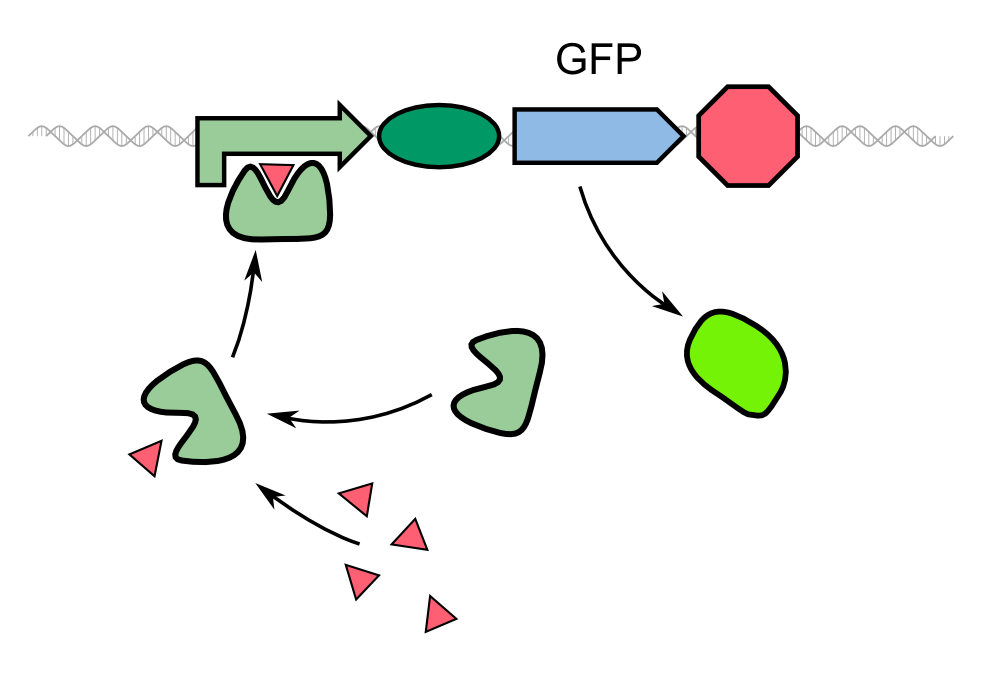
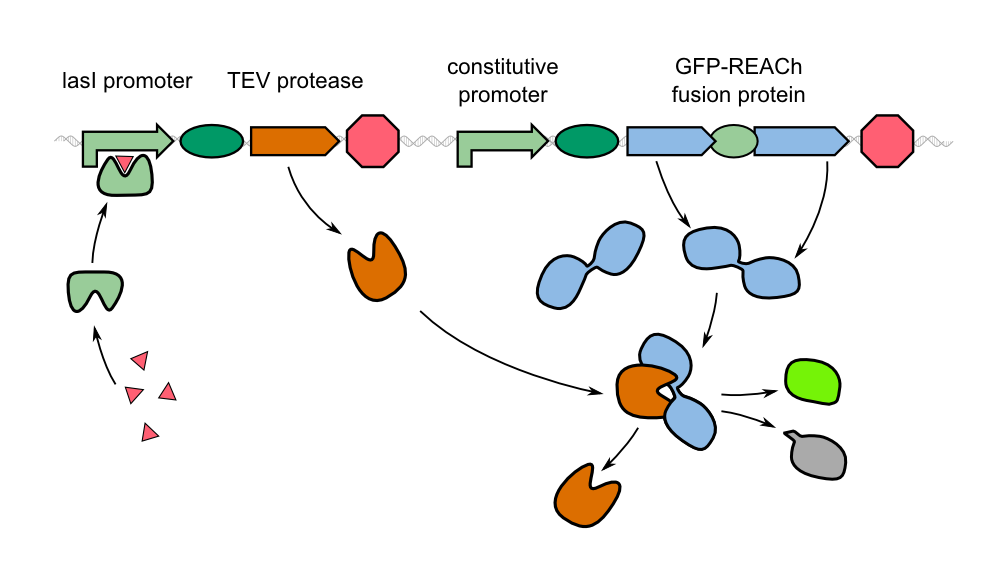
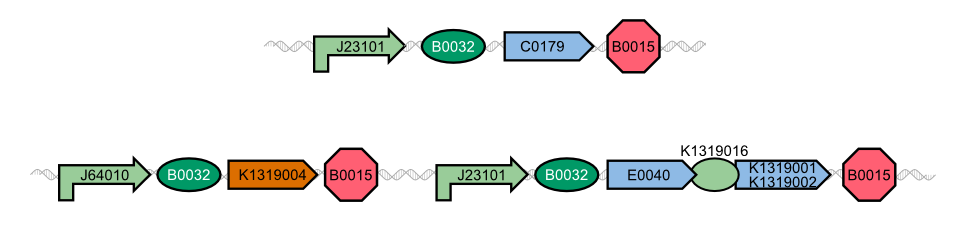
| - | In our project, we reproduced the REACh1 and REACh2 proteins by subjecting an RFC-25 compatible version of the BioBrick [http://parts.igem.org/Part:BBa_E0030 E0030] (EYFP) to a '''QuikChange mutation''', creating the BioBricks [http://parts.igem.org/Part:BBa_K1319001 K1319001] and [http://parts.igem.org/Part:BBa_K1319002 K1319002]. Subsequently, we fused each REACh protein with '''GFP (mut3b)''' which is available as BioBrick [http://parts.igem.org/Part:BBa_E0040 E0040]. The protein complex was linked via a '''protease cleavage site''' | + | In our project, we reproduced the REACh1 and REACh2 proteins by subjecting an RFC-25 compatible version of the BioBrick [http://parts.igem.org/Part:BBa_E0030 E0030] (EYFP) to a '''QuikChange mutation''', creating the BioBricks [http://parts.igem.org/Part:BBa_K1319001 K1319001] and [http://parts.igem.org/Part:BBa_K1319002 K1319002]. Subsequently, we fused each REACh protein with '''GFP (mut3b)''' which is available as BioBrick [http://parts.igem.org/Part:BBa_E0040 E0040]. The protein complex was linked via a '''protease cleavage site''' ([http://parts.igem.org/Part:BBa_K1319016 K1319016]). As constitutive promoter we use [http://parts.igem.org/Part:BBa_J23101 J232101]. When GFP is connected to either REACh quencher, GFP will absorb light but the energy will be transferred to REACh via FRET and is then emitted in the form of heat; the fluorescence is quenched. Our cells also constitutively express the '''[http://parts.igem.org/Part:BBa_C0179 LasR] activator'''. Together with the HSL molecules from ''P. aeruginosa'', LasR binds to the '''[http://parts.igem.org/Part:BBa_J64010 LasI] promoter''' that controls the expression of the TEV protease, which we made available as [http://parts.igem.org/Part:BBa_K1319004 K1319004]. When the fusion protein is cleaved by the TEV protease, REACh will be separated from GFP. The latter will then be able to absorb and emit light as usual. |
{{Team:Aachen/Figure|align=center|Aachen_14-10-08_REACh_approach_with_BioBricks_iNB.png|title= Composition of our biosensor|subtitle=For our biosensor, we use a mix of already available and self-constructed BioBricks.|width=900px}} | {{Team:Aachen/Figure|align=center|Aachen_14-10-08_REACh_approach_with_BioBricks_iNB.png|title= Composition of our biosensor|subtitle=For our biosensor, we use a mix of already available and self-constructed BioBricks.|width=900px}} | ||
| - | The resulting fusion proteins | + | The resulting fusion proteins can be expressed in [http://parts.igem.org/Part:BBa_K1319013 K1319013] (GFP fused with REACh1) and [http://parts.igem.org/Part:BBa_K1319014 K1319014] (GFP fused with REACh2). The linker between the proteins containing a TEV protease cleavage site is labelled as [http://parts.igem.org/Part:BBa_K1319016 K1319016]. |
| - | Since we did not have enough time to | + | Since we did not have enough time to build the complete system we tested our FRET reporter with an IPTG inducible TEV protease ([http://parts.igem.org/Part:BBa_K1319008 K1319008]). In this way we were able to establish a proof of concept of our reporter system including proper expression of the TEV protease as well as functionality of our GFP-REACh construct. |
{{Team:Aachen/Figure|align=center|Aachen 14-10-17 IPTG REACh iFG.png|title= Composition of our biosensor for IPTG|subtitle=To test the funtionality of our sensor concept, we expressed the TEV protease with a T7 promoter.|width=900px}} | {{Team:Aachen/Figure|align=center|Aachen 14-10-17 IPTG REACh iFG.png|title= Composition of our biosensor for IPTG|subtitle=To test the funtionality of our sensor concept, we expressed the TEV protease with a T7 promoter.|width=900px}} | ||
| + | |||
{{Team:Aachen/BlockSeparator}} | {{Team:Aachen/BlockSeparator}} | ||
| Line 166: | Line 167: | ||
<span class="anchor" id="tevprotease"></span> | <span class="anchor" id="tevprotease"></span> | ||
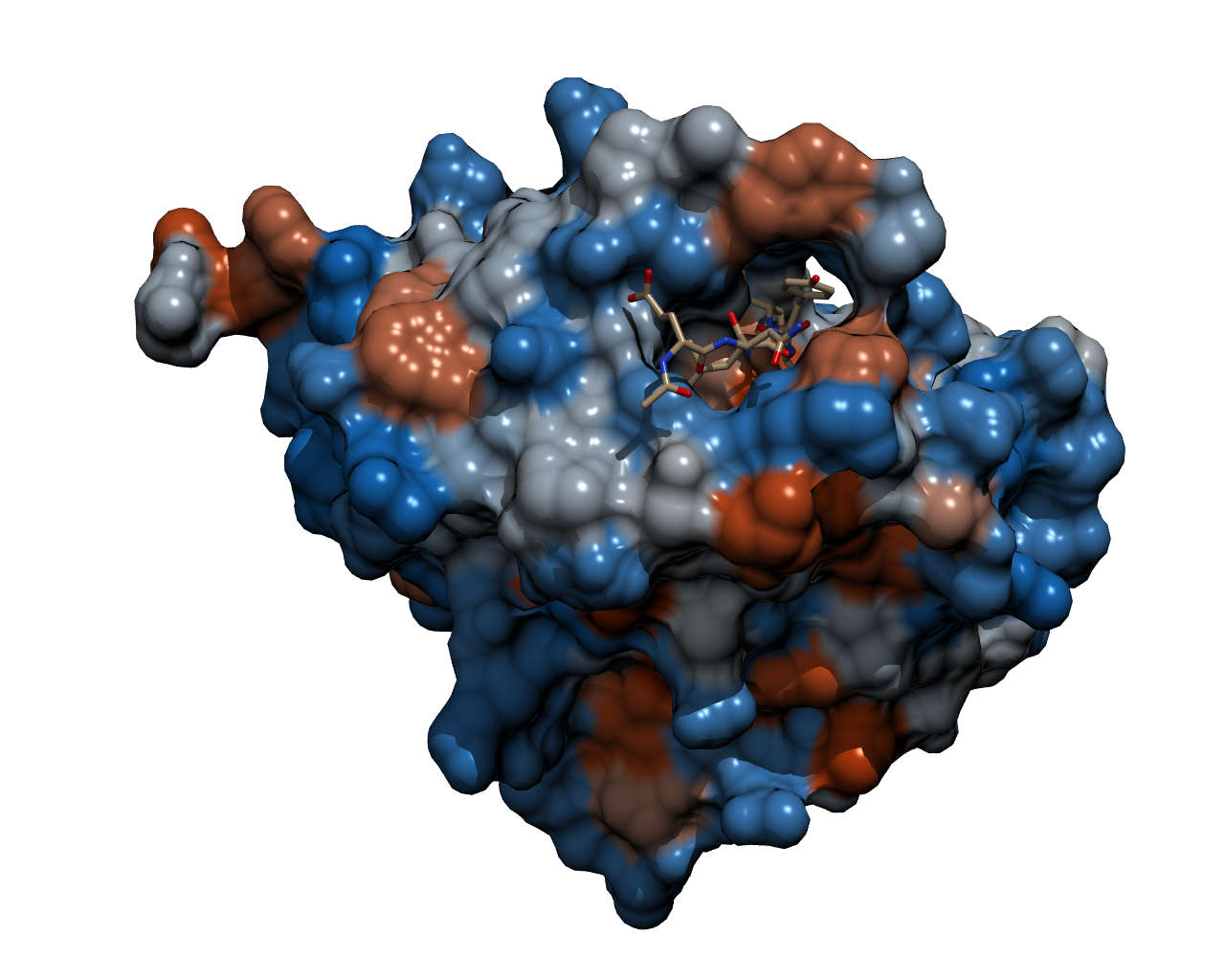
| - | To | + | To cleave the GFP-REACh fusion protein, we chose '''Tobacco Etch Virus (TEV) protease''', a highly sequence-specific cysteine protease, that is frequently used for the controlled cleavage of fusion proteins ''in vitro'' and ''in vivo''. The native protease also contains an internal self-cleavage site. This site is slowly cleaved to inactivate the enzyme. The physiological reason for the self-cleavage is unknown, however, undesired for our use. Therefore, our team uses a variant of the native TEV protease containing the mutation S219V which results in an alteration of the cleavage site so that self-inactivation is diminished. |
| - | {{Team:Aachen/Figure|Aachen_TEV_Protease_Model.png|title=TEV protease with a bound peptide|subtitle=This picture shows the TEV protease with a peptide chain bound in the binding pocket ready to be cleaved. The bound peptide chain | + | {{Team:Aachen/Figure|Aachen_TEV_Protease_Model.png|title=TEV protease with a bound peptide|subtitle=This picture shows the TEV protease with a peptide chain bound in the binding pocket ready to be cleaved. The recognition site of the bound peptide chain is located inside the binding pocket of the TEV protease. It was rendered with POV-Ray.|width=800px}} |
| - | Though quite popular in molecular biology, the TEV protease is not avaiable as a BioBrick yet. Hence, the | + | Though quite popular in molecular biology, the TEV protease is not avaiable as a BioBrick yet. Hence, the team Aachen introduces the protease with anti-self cleavage mutation S219V and codon optimized for ''E. coli'' [http://parts.igem.org/Part:BBa_K1319004 '''to the Parts Registry this year.'''] |
| Line 190: | Line 191: | ||
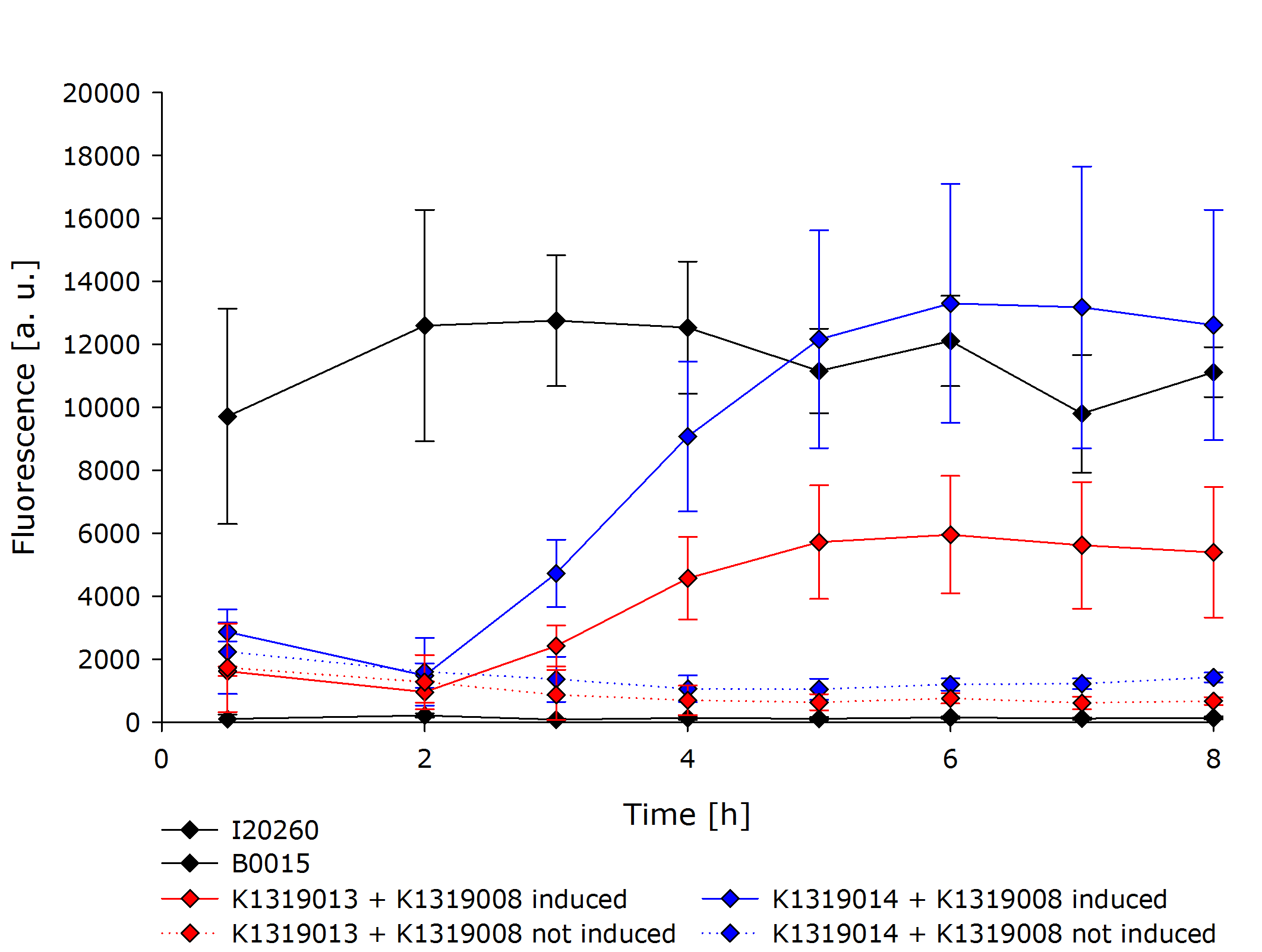
To better evaluate the fluorescence, the observed Optical desity (OD) was taken into account in order to achieve a fluorescence measurement independent of the amount of cells present. This way, the measurement represents the amount of fluorescence per cell only. | To better evaluate the fluorescence, the observed Optical desity (OD) was taken into account in order to achieve a fluorescence measurement independent of the amount of cells present. This way, the measurement represents the amount of fluorescence per cell only. | ||
| - | {{Team:Aachen/Figure|Aachen 16-10-14 Graph2iFG.PNG|title=Comparison of K1319013 + K1319008, K1319014 + K1319008, I20260 (positive control) and B0015 (negative control)|subtitle=Both double plasmid construct exhibit a clear fluorescence signal when induced.|width= | + | {{Team:Aachen/Figure|Aachen 16-10-14 Graph2iFG.PNG|title=Comparison of K1319013 + K1319008, K1319014 + K1319008, I20260 (positive control) and B0015 (negative control)|subtitle=Both double plasmid construct exhibit a clear fluorescence signal when induced.|width=700px}} |
The negative control B0015 did not exhibit any significant fluorescence. The positive control I20260 showed a steady level of fluorescence as expected due to the constitutive expression of GFP. As expected, the production is also independent of addition of IPTG therefore the triplicates have been merged together. | The negative control B0015 did not exhibit any significant fluorescence. The positive control I20260 showed a steady level of fluorescence as expected due to the constitutive expression of GFP. As expected, the production is also independent of addition of IPTG therefore the triplicates have been merged together. | ||
| Line 209: | Line 210: | ||
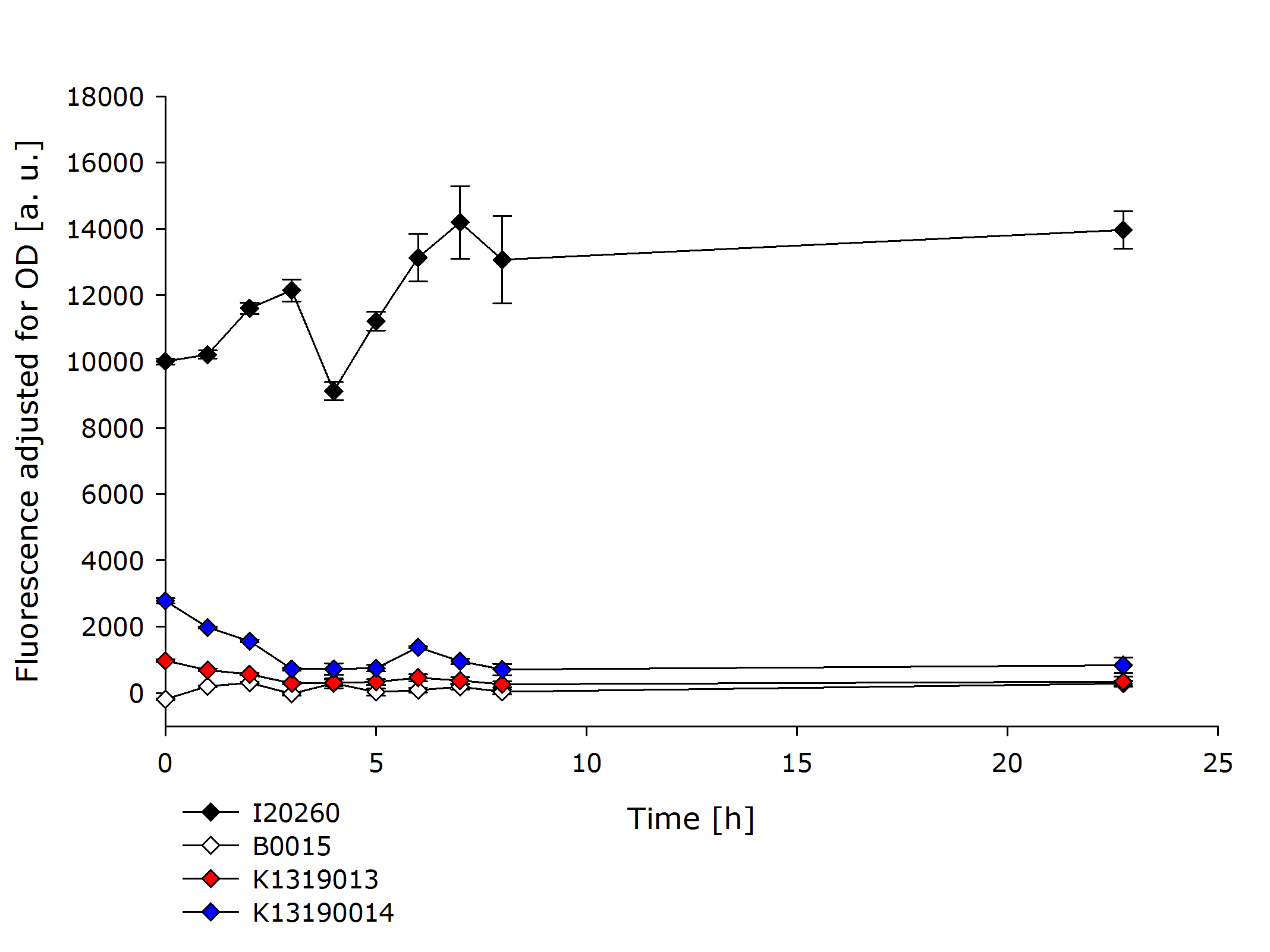
In order to further evaluate the quenching ability of the REACh1 and REACh2 constructs in the fusion proteins produced by K1319013 and K1319014, they were expressed alone without an IPTG inducible TEV protease. This eliminated the effect of a potential leakiness of the non induced promoter to reliably assess the quenching ability of the REACh1 and REACh2 proteins. | In order to further evaluate the quenching ability of the REACh1 and REACh2 constructs in the fusion proteins produced by K1319013 and K1319014, they were expressed alone without an IPTG inducible TEV protease. This eliminated the effect of a potential leakiness of the non induced promoter to reliably assess the quenching ability of the REACh1 and REACh2 proteins. | ||
| - | {{Team:Aachen/Figure|Aachen_K1319001_and_K1319002.PNG|title=Comparison of K1319013 and K1319014 with I20260 and B0015|subtitle=K1319013 and K1319014 show a severely reduced fluorescence compared to the positive control I20260.|width= | + | {{Team:Aachen/Figure|Aachen_K1319001_and_K1319002.PNG|title=Comparison of K1319013 and K1319014 with I20260 and B0015|subtitle=K1319013 and K1319014 show a severely reduced fluorescence compared to the positive control I20260.|width=700px}} |
In the previous experiment it was established that the fusion proteins K1319013 and K1319014 are expressed funtionally. K1319014 reached the same level of fluorescence as the positive control after being cut by the TEV protease. Therefore the reduced fluorescence in this experiment is completely attributable to the quenching of REACh1. The quenching reduces the fluorescence of GFP by a factor ~ 25 which means a '''quenching efficiency of ~ 96%!'''. | In the previous experiment it was established that the fusion proteins K1319013 and K1319014 are expressed funtionally. K1319014 reached the same level of fluorescence as the positive control after being cut by the TEV protease. Therefore the reduced fluorescence in this experiment is completely attributable to the quenching of REACh1. The quenching reduces the fluorescence of GFP by a factor ~ 25 which means a '''quenching efficiency of ~ 96%!'''. | ||
| Line 254: | Line 255: | ||
|<html> <img src="https://static.igem.org/mediawiki/2014/1/1a/Aachen_K13%2B8%2CK14%2B8%2CK731_slower_reduced.gif" width="480px"></html> | |<html> <img src="https://static.igem.org/mediawiki/2014/1/1a/Aachen_K13%2B8%2CK14%2B8%2CK731_slower_reduced.gif" width="480px"></html> | ||
|- | |- | ||
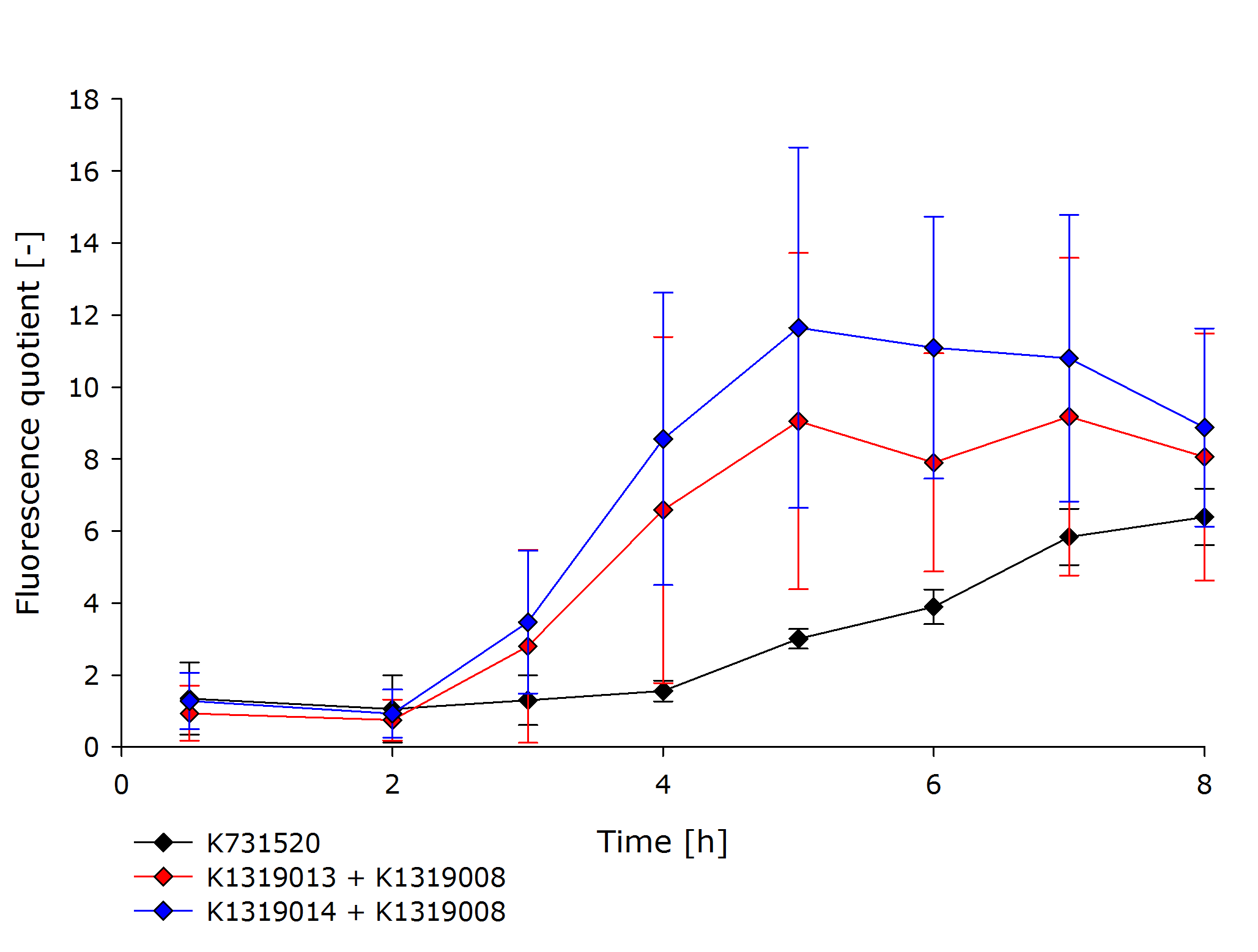
| - | |'''{{{title|K1319013 + K1319008, K1319014 + K1319008 and K731520 in an non-induced (top) and induced (bottom) chip}}}'''<br />{{{subtitle|Comparing the factor of fluorescence | + | |'''{{{title|K1319013 + K1319008, K1319014 + K1319008 and K731520 in an non-induced (top) and induced (bottom) chip}}}'''<br />{{{subtitle|Comparing the factor of fluorescence increase of induced (bottom) and not induced (top) sensor chips to the first frame of the measurement. The tested constructs are K1319013 + K1319008, K1319014 + K1319008 and K731520. After five hours, the fluorescence in our dual-plasmid REACh construct contaning K1319014 had increased by a factor of about 8, while K731520 fluorescence increased by only a factor of 2-3.}}} |
|} | |} | ||
</div> | </div> | ||
Latest revision as of 03:47, 18 October 2014
|
|
|
|
|
|
|
|
 "
"