Team:Austin Texas
From 2014.igem.org
(Difference between revisions)
Jordanmonk (Talk | contribs) m |
|||
| (29 intermediate revisions not shown) | |||
| Line 77: | Line 77: | ||
| [[Image:Austin_Texas_Measurement_Kit_Card.png|link=Team:Austin_Texas/kit]] | | [[Image:Austin_Texas_Measurement_Kit_Card.png|link=Team:Austin_Texas/kit]] | ||
| | | | ||
| - | <h1>Project: Expanded Genetic Code Measurement Kit</h1> | + | <h1>Project: Expanded Genetic Code Measurement Kit</h1><div align=justify> |
| - | + | Noncanonical amino acids (ncAAs) are an exciting new tool in the synthetic biologist's toolbox. Incorporating ncAAs into proteins will allow scientists to create bacteria that can perform novel functions. Unfortunately, these ncAAs are often difficult to effectively employ due to poor characterization of the synthetase/tRNA pair, which may mean that the pair have low fidelity or low efficiency. Our project aims to create a cheap, easy-to-use universal kit that can quickly measure the fidelity and incorporation efficiency of any synthetase/tRNA pair. The kit is a simple two-plasmid system. The first plasmid contains an IPTG-inducible RFP-sfGFP reporter. The two domains are connected by a linker sequence containing either a tyrosine codon (as a control) or a recoded amber stop codon (where the ncAA will be incorporated). The other plasmid contains the synthetase/tRNA pair. After the RFP-linker-GFP protein is expressed, the fluorescence of each fluorescent domain of the fusion protein is measured and compared. Depending on the relative intensities of the RFP and GFP fluorescence under different conditions, we can determine both the fidelity of the synthetase/tRNA pair as well as the efficiency of the pair at incorporating the ncAA. '''With this quick and easy “plug and play” system, researchers will be able to insert any plasmid containing a new synthetase/tRNA pair into our Expanded Genetic Code Measurement kit to quickly characterize the synthetase/tRNA pair.'''</div> | |
| - | Noncanonical amino acids (ncAAs) are an exciting new tool in the | + | |
|- valign="top" | |- valign="top" | ||
| [[Image:Austin_Texas_Photocage_Card.png|link=Team:Austin_Texas/photocage]] | | [[Image:Austin_Texas_Photocage_Card.png|link=Team:Austin_Texas/photocage]] | ||
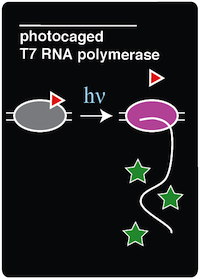
| - | | <h1>Project: Photocaged T7 RNA Polymerase</h1> | + | | <h1>Project: Photocaged T7 RNA Polymerase</h1><div align=justify> |
| + | Noncanonical amino acids (ncAAs) may have unique chemical groups that can provide very useful functionality not normally present in an organism. This summer we used a photocaged ncAA, ortho-nitrobenzyl tyrosine (ONBY), to prevent protein functionality until spatially and/or temporally triggered by light. '''To this end, our team successfully recreated a light-activatable T7 RNA Polymerase (RNAP) for a method of noninvasive, spaciotemporal control of protein expression.''' We chose T7 RNAP because there is a tyrosine residue in the active site of the polymerase. By changing this codon to an amber codon, we were able to incorporate (ONBY), a photocaged tyrosine, into the active site. The presence of the extra ONB group inhibits T7 RNAP activity. When exposed to 365 nm light, the molecular cage (the ONB group) is released, leaving tyrosine. Thus, T7 RNAP is only functional when exposed to 365 light, which restores the native T7 polymerase active site that is necessary for proper function of the enzyme. To test this system, we used a GFP reporter with a T7 promoter. '''We were able to see a significant increase in GFP expression upon photodecaging T7 RNAP, as expected.'''</div> | ||
| + | |- valign="top" | ||
| + | | [[file:caffeinecolisxsw.png|200px| link=https://2014.igem.org/Team:Austin_Texas/human_practices]] | ||
| + | |<h1>Human Practices: Caffeinated Coli @ SXSW Create</h1><div align=justify> | ||
| + | In the spring of 2014, the UT iGEM team was invited to create a booth at the [http://sxsw.com/exhibitions/sxsw-create/participants-2014 SXSW Interactive Create Event.] Team members took the opportunity to educate the public on the applications and potential benefits of genetically modified organisms. In a concerted effort between past and present team members, the group banded together for the first time to create an informative, and fun presentation relating to the previous iGEM 2012 team project, [https://2012.igem.org/Team:Austin_Texas/Caffeinated_coli| ''Caffeinated Coli.''] '''Our new project also went further scientifically, using Caffeinated Coli to calculate the amount of caffeine found in coffee, an organic beverage that is not formulaic.''' This allowed us to reach out to the wider Austin community and talk with them about science and synthetic biology by using something that many of them consume on a daily basis.</div> | ||
| + | |} | ||
| - | + | {| width="100%" cellpadding=10 | |
| - | + | |- | |
| - | + | | rowspan="2" | [[Image:UT_Austin_CSSB.png|link=http://cssb.utexas.edu]] | |
| - | |- | + | | rowspan="2" | [[Image:UT_Austin_BEACON.png|180px|link=http://beacon-center.org]] |
| - | | | + | | [[Image:UT_Austin_ICMB.png|link=http://icmb.utexas.edu|400px]] |
| - | | | + | |- |
| - | + | | [[Image:UT_Austin_MBS.png|link=http://molecularbiosci.utexas.edu|400px]] | |
|} | |} | ||
Latest revision as of 03:55, 18 October 2014
| ||||||||||||||||||||||||||||||||||||||
 "
"