Team:Heidelberg/pages/Xylanase
From 2014.igem.org
Contents |
Introduction
Endo-1, 4-β-xylanases (in short Xylanases) are enzymes that hydrolyze the internal glycosidic bonds within the xylan backbone. Xylan is one major components of wood (accounting for approximately 20-35% of plant cell wall dry mass) and therefore one of the most abundant organic molecules in nature [1]. A variety of prokaryotes and eukaryotes, but not animals, express Xylanases to use the monosaccharides from Xylan. We used a recombinant XylA from Bacillus subtilis expressed in E. coli because it is a well-established model organism and the XylA because of its structural properties. Xylanases find broad application in industry, especially for bio-bleaching in pulp production, animal feedstocks and the production of bio-Ethanol. For these applications often more stable variants are needed and therefore the engineering of heat stable xylanases is an active research topic. Common approaches to this problem are to investigate potentially heat-stable xylanases fromthermophilic archae or bacteria and to introduce disulfide bonds in order to generate more robust xylanases.
We want to introduce intein-mediated circularization as an alternative approach to confer heat stabilityproteins. We chose the xylanase of Bacillus subtilis which is a promising enzyme to be used in pulp bleaching. Moreover, N- and C-terminus of this xylanase are in close (4.6 Å , see Figure X) which facilitates circularization. To achieve circularization we used split inteins . This method produces after the splicing reaction a scar of 6 amino acids, which can be bridge the gap between the native termini.
Experimental Procedures
Design and cloning of constructs
To generate a circular, heat-stable xylanase we chose to follow two strategies and circularize not only the native form but also a xylanase including a histidine tag for purification. For circularization we used the split NpuDnaE intein, since it is used as the golden standard for protein splicing. All cloning steps were carried out using our new RFC[i] standard procedure. An overview of all constructs including controls for circularization is shown in the following Table.
| Construct Names | Motivation |
| Linear | native xylanase |
| Linear +His | control for “Circular with His6-Tag” construct |
| Linear+His+ Ext | control with similar amino acid composition as the circular His6-tag xylanase (“Circular with His-tag”) |
| Circular | native xylanase with split NpuDnaE split intein |
| Circular + His | native xylanase with split NpuDnaE split intein and His6-tag |
| Circular +His+ destr.Ext. | control with mutation in C-Ext. (Cys-> Gly) |
For the construct to express linear xylanase (BBa_K1362020) the coding sequence of xylanase without start codon and export signal was amplified from B.subtilis by PCR. Overhangs containing an EcoRI site, the RBS B0034, a start codon (forward) and a SpeI site and a PstI site (reverse) were added by primer overhangs. The PCR product was cut with X and P and ligated into the expression vector pSBX1K30. In order to create the constructs to express linear xylanase with His6-Tag BBa_K1362023 and linear xylanase with His6-tag and exteins, primers to linearize BBa_K1362020 between the xylanase coding sequence and the stop codon and add BsaI sites at the ends were designed. Inserts coding for the sequence GSHHHHHHSG (His6-Tag) respectively HHHHHHRGKCWE (His6-tag + exteins) with overhangs corresponding to the overhangs caused by BsaI restriction of the vector PCR product were designed and ordered as DNA-oligomers. The inserts were annealed and inserted into the purified PCR product of BBa_K1362020 by Golden Gate cloning.
The constructs to express circular xylanase (BBa_K1362022), circular xylanase with His6-tag (BBa_K1362023) and non-circular xylanase with His6-tag (with a Cys->Gly mutation in the C-extein) are based on the NpuDnaE intein RFC[i circularization construct]. For BBa_K1362022, the coding sequence of xylanase without start codon, export signal and stop codon was amplified from B.subtilis by PCR. Overhangs containing RFC[i] E standard overhang, BsaI sites, the coding sequences of the exteins CWE (forward) and RGK, the RFC[i] B standard overhang, BsaI (reverse) were added by primer overhangs. The PCR product was purified and it was inserted into the NpuDnaE intein RFC[i circularization construct] by Golden Gate assembly. BBa_K1362023 and the construct to express non-circular xylanase with His6-tag (with a Cys->Gly mutation in the C-extein) were cloned in the same way, except for the additional His6-tags in the primer overhangs.
Xylanase Assay
To test if circularization increases heat stability of B.subtilis xylanase we determined xylanase activity after 30min incubation at different temperatures using the EnzChek® Ultra Xylanase Assay Kit. This kit includes a fluorescently labelled hemicellulose substrate. Xylanase-mediated hydrolysis of the substrate results in the unquenching of the attached fluorescent dyes and thus increased fluorescence. Fluorescence intensity over time was measured using the Tecan Infinite M200 reader and xylanase from Thermomyces lanuginosus was used as an initial positive control. Data were analyzed and visualized using a python script similar to the one for the lysozyme assay.
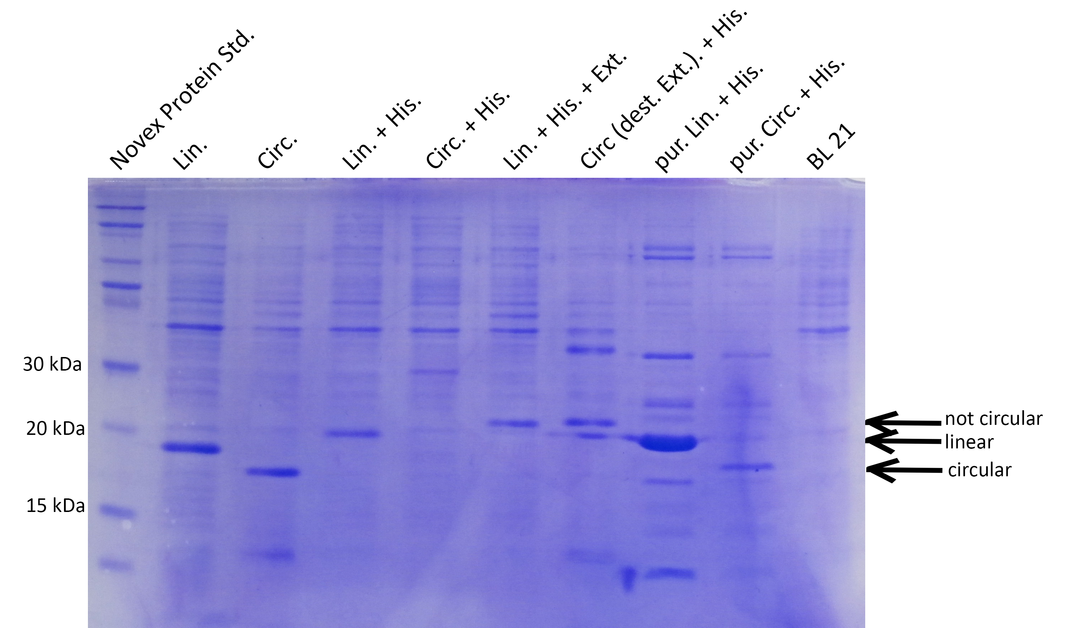
Results
1) Expression of linear and circular Xylanase
To generate a circular, heat-stable xylanase we chose to follow two strategies and circularize not only the native form but also a His6-tagged xylanase. His-tagged xylanase can be easily purified and thus, xylanase activity can be determined more reliably and without interfering molecules that are otherwise present in the lysate. On the other hand, the His-tag might influence thermostability and xylanase activity. Therefore we also included the unmodified xylanase and measured activity directly in bacterial lysate similar to the lysozyme assay.
For circularization we used the split NpuDnaE intein and performed cloning using our new RFC[i] standard procedure.
 "
"